当前位置:
X-MOL 学术
›
Org. Lett.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Strategy for Overcoming Full Reversibility of Intermolecular Radical Addition to Aldehydes: Tandem C–H and C–O Bonds Cleaving Cyclization of (Phenoxymethyl)arenes with Carbonyls to Benzofurans
Organic Letters ( IF 4.9 ) Pub Date : 2018-05-18 00:00:00 , DOI: 10.1021/acs.orglett.8b01207 Hong-Xing Zheng 1 , Xiang-Huan Shan 1 , Jian-Ping Qu 2 , Yan-Biao Kang 1
Organic Letters ( IF 4.9 ) Pub Date : 2018-05-18 00:00:00 , DOI: 10.1021/acs.orglett.8b01207 Hong-Xing Zheng 1 , Xiang-Huan Shan 1 , Jian-Ping Qu 2 , Yan-Biao Kang 1
Affiliation
|
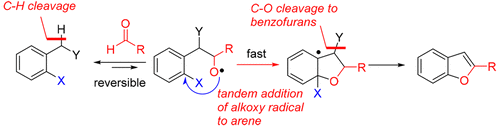
An intermolecular addition of carbon radicals enabled by a cascade radical coupling strategy is developed. It includes an intermolecular alkyl radical addition to a carbonyl group followed by an intramolecular alkoxy radical addition to haloarenes and produces substituted benzofurans in high yields. The radical nature of this reaction is explored by radical trapping experiments and EPR analysis. The mechanism is investigated by KIE experiments and control experiments. This method could provide rapid and practical access to the key intermediate of TAM-16, a safe and potent antibacterial agent for treating tuberculosis, and, therefore, is of great importance for organic synthesis and the pharmaceutical industry.
中文翻译:

克服分子间自由基加成醛的完全可逆性的策略:串联的C–H和C–O键使(苯氧基甲基)芳烃与羰基的环化裂解为苯并呋喃
通过级联自由基偶联策略实现了碳自由基的分子间加成。它包括向羰基的分子间烷基加成,然后向卤代芳烃的分子内烷氧基加成,并以高收率产生取代的苯并呋喃。通过自由基捕获实验和EPR分析,探索了该反应的自由基性质。通过KIE实验和控制实验研究了该机理。该方法可以快速,实用地获得TAM-16的关键中间体,TAM-16是治疗结核病的安全有效的抗菌剂,因此,对有机合成和制药工业具有重要意义。
更新日期:2018-05-18
中文翻译:

克服分子间自由基加成醛的完全可逆性的策略:串联的C–H和C–O键使(苯氧基甲基)芳烃与羰基的环化裂解为苯并呋喃
通过级联自由基偶联策略实现了碳自由基的分子间加成。它包括向羰基的分子间烷基加成,然后向卤代芳烃的分子内烷氧基加成,并以高收率产生取代的苯并呋喃。通过自由基捕获实验和EPR分析,探索了该反应的自由基性质。通过KIE实验和控制实验研究了该机理。该方法可以快速,实用地获得TAM-16的关键中间体,TAM-16是治疗结核病的安全有效的抗菌剂,因此,对有机合成和制药工业具有重要意义。











































 京公网安备 11010802027423号
京公网安备 11010802027423号