Chem ( IF 23.5 ) Pub Date : 2018-05-17 , DOI: 10.1016/j.chempr.2018.04.008 Jichao Xiao , Yuli He , Feng Ye , Shaolin Zhu
|
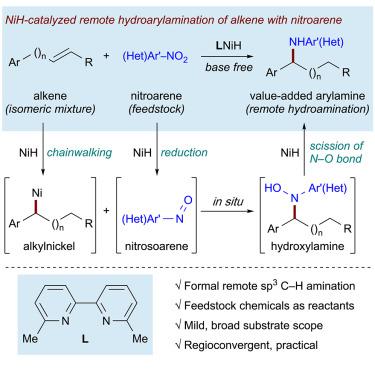
Direct installation of a functional group at remote, unfunctionalized sites in an alkyl chain is a synthetically valuable but rarely reported process. The remote relay hydroarylamination of distal and proximal olefins, and of olefin isomeric mixtures, has been achieved through NiH-catalyzed alkene isomerization and sequential reductive hydroarylamination with nitroarenes. This provides an attractive approach to the direct installation of a distal arylamino group within alkyl chains. The single-step conversion of simple olefins and nitro(hetero)arenes to value-added arylamines is a practical strategy for amine synthesis as well as the remote activation of sp3 C–H bonds. The value of this transformation is further supported by the regioconvergent arylamination of isomeric mixtures of olefins.
中文翻译:

硝基芳烃对烯烃的远程sp 3 C–H胺化反应
将官能团直接安装在烷基链中偏远未官能化的位置上是具有合成价值的,但鲜有报道。远端和近端烯烃以及烯烃异构体混合物的远程中继加氢芳基化已通过NiH催化的烯烃异构化和与硝基芳烃的顺序还原加氢芳基化实现。这为将远端的芳基氨基直接安装在烷基链中提供了一种有吸引力的方法。从简单的烯烃和硝基(杂)芳烃到增值芳胺的单步转化是胺合成以及sp 3 C–H键的远程活化的一种实用策略。烯烃异构体混合物的区域会聚芳基化进一步支持了这种转化的价值。



























 京公网安备 11010802027423号
京公网安备 11010802027423号