Bioorganic Chemistry ( IF 5.1 ) Pub Date : 2018-05-15 , DOI: 10.1016/j.bioorg.2018.05.004 Mohammad Shahidul Islam , Assem Barakat , Abdullah Mohammed Al-Majid , M. Ali , Sammer Yousuf , M. Iqbal Choudhary , Ruqaiya Khalil , Zaheer Ul-Haq
|
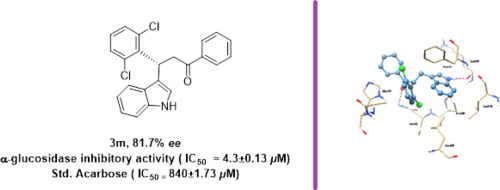
Indole containing compounds have acquired conspicuous significance due to their wide spectrum of biological activities. Synthesis of a series of enantiomerically pure indole derivatives 3a-r via Friedel–Crafts alkylation of indole 1 with enones 2a-r were described here. The products were isolated in a moderate to excellent yields (upto 89%) with excellent enantioselectivities (upto 99.9% ee). These compounds 3a-r were evaluated for their in vitro α-glucosidase inhibitory activity and some of them were identified as potent inhibitors (IC50 = 4.3 ± 0.13–43.9 ± 0.51 μM) with several fold higher activity than the clinically used α-glucosidase inhibitor, acarbose (IC50 = 840 ± 1.73 μM). To the best of knowledge, this is the first report of the propanone substituted indole ring containing compounds by in vitro α-glucosidase enzyme inhibition.
中文翻译:

吲哚衍生物是新颖的催化不对称合成α葡糖苷酶抑制剂在体外
含吲哚的化合物由于其广泛的生物活性而具有显着的意义。一系列对映体纯的吲哚衍生物的合成图3a - - [R 通过吲哚的Friedel-Crafts烷基化1与烯酮图2a-R这里进行了描述。以中等至优异的产率(高达89%)和优异的对映选择性(高达99.9%ee)分离产物。这些化合物3A - - [R为他们进行了评价体外α葡萄糖苷酶抑制活性,并且它们中的一些被确定为有效的抑制剂(IC 50 = 4.3±0.13-43.9±0.51 μM)与几个比临床使用的倍高的活性α葡糖苷酶抑制剂阿卡波糖(IC 50 = 840±1.73 μ M)。就其所知,这是首次报道了通过体外α-葡萄糖苷酶抑制作用,含有丙酮取代的吲哚环的化合物。



























 京公网安备 11010802027423号
京公网安备 11010802027423号