European Journal of Medicinal Chemistry ( IF 6.0 ) Pub Date : 2018-05-11 , DOI: 10.1016/j.ejmech.2018.04.056 Bruno Oyallon , Marie Brachet-Botineau , Cédric Logé , Pascal Bonnet , Mohamed Souab , Thomas Robert , Sandrine Ruchaud , Stéphane Bach , Pascal Berthelot , Fabrice Gouilleux , Marie-Claude Viaud-Massuard , Caroline Denevault-Sabourin
|
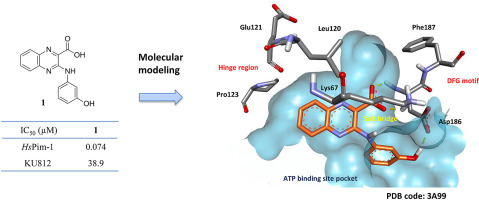
We identified a new series of quinoxaline-2-carboxylic acid derivatives, targeting the human proviral integration site for Moloney murine leukemia virus-1 (HsPim-1) kinase. Seventeen analogues were synthesized providing useful insight into structure-activity relationships studied. Docking studies realized in the ATP pocket of HsPim-1 are consistent with an unclassical binding mode of these inhibitors. The lead compound 1 was able to block HsPim-1 enzymatic activity at nanomolar concentrations (IC50 of 74 nM), with a good selectivity profile against a panel of mammalian protein kinases. In vitro studies on the human chronic myeloid leukemia cell line KU812 showed an antitumor activity at micromolar concentrations. As a result, compound 1 represents a promising lead for the design of novel anticancer targeted therapies.
中文翻译:

新型喹喔啉-2-羧酸及其类似物作为Pim-1抑制剂的结构设计
我们确定了一系列新的喹喔啉-2-羧酸衍生物,针对莫洛尼鼠白血病病毒1(Hs Pim-1)激酶的人类前病毒整合位点。合成了十七个类似物,为研究结构-活性关系提供了有用的见识。在Hs Pim-1的ATP口袋中实现的对接研究与这些抑制剂的非经典结合模式一致。铅化合物1能够在纳摩尔浓度(IC 50为74 nM)下阻断Hs Pim-1的酶促活性,对一组哺乳动物蛋白激酶具有良好的选择性。体外对人类慢性髓样白血病细胞株KU812的研究显示,在微摩尔浓度下具有抗肿瘤活性。结果,化合物1代表了设计新型抗癌靶向疗法的有希望的先导。









































 京公网安备 11010802027423号
京公网安备 11010802027423号