当前位置:
X-MOL 学术
›
ACS Catal.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Cross-Coupling of Phenol Derivatives with Umpolung Aldehydes Catalyzed by Nickel
ACS Catalysis ( IF 11.3 ) Pub Date : 2018-04-17 00:00:00 , DOI: 10.1021/acscatal.8b01224 Leiyang Lv 1, 2 , Dianhu Zhu 1 , Jianting Tang 1 , Zihang Qiu 1 , Chen-Chen Li 1 , Jian Gao 1 , Chao-Jun Li 1
ACS Catalysis ( IF 11.3 ) Pub Date : 2018-04-17 00:00:00 , DOI: 10.1021/acscatal.8b01224 Leiyang Lv 1, 2 , Dianhu Zhu 1 , Jianting Tang 1 , Zihang Qiu 1 , Chen-Chen Li 1 , Jian Gao 1 , Chao-Jun Li 1
Affiliation
|
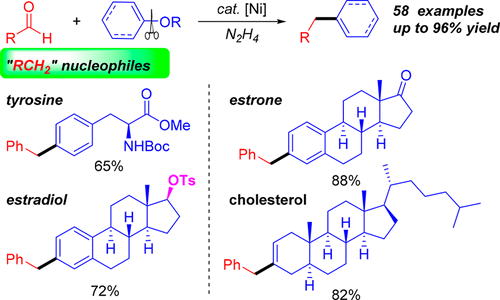
A nickel-catalyzed cross-coupling to construct the C(sp2)–C(sp3) bond was developed from two sustainable biomass-based feedstocks: phenol derivatives with umpolung aldehydes. This strategy features the in situ generation of moisture/air-stable hydrazones from naturally abundant aldehydes, which act as alkyl nucleophiles under catalysis to couple with readily available phenol derivatives. The avoidance of using both halides as the electrophiles and organometallic or organoboron reagents (also derived from halides) as the nucleophiles makes this method more sustainable. Water tolerance, great functional group (ketone, ester, free amine, amide, etc.) compatibility, and late-stage elaboration of complex biological molecules exemplified its practicability and unique chemoselectivity over organometallic reagents.
中文翻译:

镍催化的酚衍生物与with醛的交叉偶联
镍催化交叉偶联以构建C(sp 2)–C(sp 3)债券是从两种可持续的以生物质为基础的原料开发出来的:苯酚衍生物和戊醛。该策略的特征是由天然丰富的醛原位生成水分/空气不稳定的azo,这些醛在催化下可与烷基亲核试剂偶联,并与易得的苯酚衍生物偶联。避免同时使用卤化物作为亲电子试剂和避免使用有机金属或有机硼试剂(也衍生自卤化物)作为亲核试剂使该方法更具可持续性。耐水性,强大的官能团(酮,酯,游离胺,酰胺等)的相容性,以及复杂生物分子的后期精制,证明了其与有机金属试剂相比具有实用性和独特的化学选择性。
更新日期:2018-04-17
中文翻译:

镍催化的酚衍生物与with醛的交叉偶联
镍催化交叉偶联以构建C(sp 2)–C(sp 3)债券是从两种可持续的以生物质为基础的原料开发出来的:苯酚衍生物和戊醛。该策略的特征是由天然丰富的醛原位生成水分/空气不稳定的azo,这些醛在催化下可与烷基亲核试剂偶联,并与易得的苯酚衍生物偶联。避免同时使用卤化物作为亲电子试剂和避免使用有机金属或有机硼试剂(也衍生自卤化物)作为亲核试剂使该方法更具可持续性。耐水性,强大的官能团(酮,酯,游离胺,酰胺等)的相容性,以及复杂生物分子的后期精制,证明了其与有机金属试剂相比具有实用性和独特的化学选择性。











































 京公网安备 11010802027423号
京公网安备 11010802027423号