当前位置:
X-MOL 学术
›
ACS Chem. Biol.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Capturing Post-Translational Modification-Triggered Protein–Protein Interactions Using Dual Noncanonical Amino Acid Mutagenesis
ACS Chemical Biology ( IF 3.5 ) Pub Date : 2018-03-15 00:00:00 , DOI: 10.1021/acschembio.8b00021 Yunan Zheng 1 , Martin J. Gilgenast 1 , Sacha Hauc 1 , Abhishek Chatterjee 1
ACS Chemical Biology ( IF 3.5 ) Pub Date : 2018-03-15 00:00:00 , DOI: 10.1021/acschembio.8b00021 Yunan Zheng 1 , Martin J. Gilgenast 1 , Sacha Hauc 1 , Abhishek Chatterjee 1
Affiliation
|
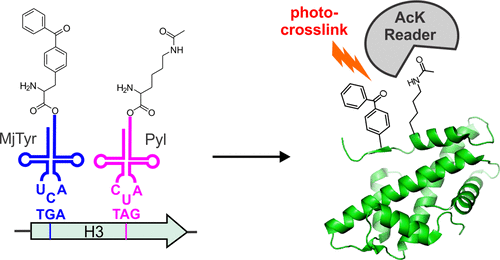
Reversible post-translational modification (PTM) is a powerful and ubiquitous mechanism to regulate protein function. The mechanistic basis of the associated functional regulation by PTMs often involves the recruitment of interaction partners that selectively bind the modified protein. Identifying such functionally important protein–protein interactions that are uniquely triggered by PTMs remains difficult due to several technical challenges. To address this, here we develop technology to site-specifically incorporate two distinct noncanonical amino acids into recombinant proteins: one modeling a PTM of interest and the second harboring a photoaffinity probe. Using lysine-23 acetylation of histone 3 as a model system, we show that such dual-labeled “protein probes” can covalently capture its “reader” protein.
中文翻译:

使用双重非典型氨基酸诱变捕获翻译后修饰触发的蛋白质-蛋白质相互作用
可逆翻译后修饰(PTM)是调节蛋白质功能的强大且普遍存在的机制。PTM进行相关功能调节的机制基础通常涉及募集选择性结合修饰蛋白的相互作用伴侣。由于一些技术挑战,要识别由PTM独特触发的功能上重要的蛋白质之间的相互作用仍然很困难。为了解决这个问题,我们在这里开发了将两个不同的非经典氨基酸位点特异性结合到重组蛋白中的技术:一种模拟感兴趣的PTM,第二种模拟光亲和探针。使用组蛋白3的赖氨酸23乙酰化作为模型系统,我们显示出这种双重标记的“蛋白质探针”可以共价捕获其“阅读器”蛋白质。
更新日期:2018-03-15
中文翻译:

使用双重非典型氨基酸诱变捕获翻译后修饰触发的蛋白质-蛋白质相互作用
可逆翻译后修饰(PTM)是调节蛋白质功能的强大且普遍存在的机制。PTM进行相关功能调节的机制基础通常涉及募集选择性结合修饰蛋白的相互作用伴侣。由于一些技术挑战,要识别由PTM独特触发的功能上重要的蛋白质之间的相互作用仍然很困难。为了解决这个问题,我们在这里开发了将两个不同的非经典氨基酸位点特异性结合到重组蛋白中的技术:一种模拟感兴趣的PTM,第二种模拟光亲和探针。使用组蛋白3的赖氨酸23乙酰化作为模型系统,我们显示出这种双重标记的“蛋白质探针”可以共价捕获其“阅读器”蛋白质。










































 京公网安备 11010802027423号
京公网安备 11010802027423号