Bioorganic Chemistry ( IF 4.5 ) Pub Date : 2018-03-20 , DOI: 10.1016/j.bioorg.2018.03.023 Ibrahim Chaaban 1 , Ola H Rizk 2 , Tamer M Ibrahim 3 , Shery S Henen 1 , El-Sayeda M El-Khawass 1 , Aida E Bayad 4 , Ibrahim M El-Ashmawy 5 , Hisham A Nematalla 6
|
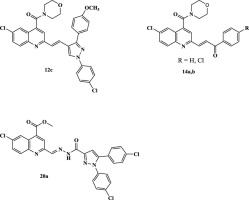
New quinoline compounds comprising pyrazole scaffold through different amide linkages were synthesized. The synthesized compounds were evaluated for their anti-inflammatory activity. Eight compounds (5c, 11b,c, 12c, 14a,b, 20a and 21a) were found to exhibit promising anti-inflammatory profiles in acute and sub-acute inflammatory models. They were screened for their ulcerogenic activity and none of them showed significant ulcerogenic activity comparable to the reference drug celecoxib and are well tolerated by experimental animals with high safety margin (ALD50 > 0.3 g/kg). Compounds 5c, 11b,c, 12c, 14a,b, 20a and 21a showed significant in vitro LOX inhibitory activity higher than that of zileuton. In vitro COX-1/COX-2 inhibition study revealed that compounds 12c, 14a,b and 20a showed higher selectivity towards COX-2 than COX-1. Among the tested compounds, 12c, 14a and 14b showed the highest inhibitory activity against COX-2 with an IC50 values of 0.1, 0.11 and 0.11 μM respectively. The docking experiments attempted to postulate the binding mode for the most active compounds in the binding site of COX-2 enzymes and confirmed the high selectivity binding towards COX-2 enzyme over COX-1.
中文翻译:

一些新型喹啉衍生物的合成、抗炎筛选、分子对接和COX-1,2/-5-LOX抑制谱
合成了通过不同酰胺键包含吡唑骨架的新喹啉化合物。评估合成的化合物的抗炎活性。八种化合物( 5c 、 11b、c 、 12c 、 14a、b 、 20a和21a )被发现在急性和亚急性炎症模型中表现出有希望的抗炎作用。对它们的致溃疡活性进行了筛选,没有一个显示出与参考药物塞来昔布相当的显着致溃疡活性,并且实验动物具有良好的耐受性,安全裕度高(ALD 50 > 0.3 g/kg)。化合物5c 、 11b、c 、 12c 、 14a、b 、 20a和21a显示出比齐留通更高的显着体外LOX抑制活性。体外COX-1/COX-2抑制研究表明,化合物12c 、 14a、b和20a对COX-2的选择性高于COX-1。在测试的化合物中, 12c 、 14a和14b对COX-2表现出最高的抑制活性,IC 50值分别为0.1、0.11和0.11μM。对接实验试图推测 COX-2 酶结合位点中最活跃的化合物的结合模式,并证实与 COX-1 相比,对 COX-2 酶的高选择性结合。











































 京公网安备 11010802027423号
京公网安备 11010802027423号