当前位置:
X-MOL 学术
›
New J. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
A possible target: triple-bonded indiumantimony molecules with high stability†
New Journal of Chemistry ( IF 2.7 ) Pub Date : 2018-03-19 00:00:00 , DOI: 10.1039/c8nj00549d Jia-Syun Lu,Ming-Chung Yang,Ming-Der Su
New Journal of Chemistry ( IF 2.7 ) Pub Date : 2018-03-19 00:00:00 , DOI: 10.1039/c8nj00549d Jia-Syun Lu,Ming-Chung Yang,Ming-Der Su

|
We have considered as a theoretical possibility the development of triple-bonded RIn![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SbR molecules bearing suitable substituents (R). Calculations have demonstrated that the RIn
SbR molecules bearing suitable substituents (R). Calculations have demonstrated that the RIn![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SbR molecules possessing smaller substituents (such as R = F, OH, CH3, H, and SiH3) cannot be stabilized. Only the triple-bonded R′In
SbR molecules possessing smaller substituents (such as R = F, OH, CH3, H, and SiH3) cannot be stabilized. Only the triple-bonded R′In![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SbR′ molecules featuring sterically bulky groups (R′ = SiMe(SitBu3)2, SiiPrDis2, Tbt (= C6H2-2,4,6-{CH(SiMe3)2}3), and Ar* (= C6H3-2,6-(C6H2-2,4,6-i-Pr3)2)) are found to locate on the global minimum of the singlet potential energy surface and are thermodynamically stable. The valence-electron bonding model reveals that the bonding nature of R′In
SbR′ molecules featuring sterically bulky groups (R′ = SiMe(SitBu3)2, SiiPrDis2, Tbt (= C6H2-2,4,6-{CH(SiMe3)2}3), and Ar* (= C6H3-2,6-(C6H2-2,4,6-i-Pr3)2)) are found to locate on the global minimum of the singlet potential energy surface and are thermodynamically stable. The valence-electron bonding model reveals that the bonding nature of R′In![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SbR′ can be represented as
SbR′ can be represented as 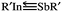 . Our computational investigations based on several theoretical methods (i.e., the charge decomposition analysis, the natural bond orbital analysis and the natural resonance theory) reveal that both the electronic and steric effects of bulkier substituent groups play important roles in making triple-bonded R′In
. Our computational investigations based on several theoretical methods (i.e., the charge decomposition analysis, the natural bond orbital analysis and the natural resonance theory) reveal that both the electronic and steric effects of bulkier substituent groups play important roles in making triple-bonded R′In![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SbR′ species synthetically accessible and isolable in a stable form.
SbR′ species synthetically accessible and isolable in a stable form.
中文翻译:

可能的目标:![[三重键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e002.gif) 具有高稳定性的三键合铟锑分子†
具有高稳定性的三键合铟锑分子†
我们已考虑开发![[三重键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e002.gif) 带有合适取代基(R)的三键RIn SbR分子作为理论上的可能性。计算表明,
带有合适取代基(R)的三键RIn SbR分子作为理论上的可能性。计算表明,![[三重键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e002.gif) 具有较小取代基(如R = F,OH,CH 3,H和SiH 3)的RIn SbR分子无法稳定。只有三键结合的R'In
具有较小取代基(如R = F,OH,CH 3,H和SiH 3)的RIn SbR分子无法稳定。只有三键结合的R'In ![[三重键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e002.gif) SbR'分子具有空间庞大的基团(R'= SiMe(Si t Bu 3)2,SiiPrDis 2,Tbt(= C 6 H 2 -2,4,6- {CH(SiMe 3)2 } 3)和Ar *(= C 6 H 3 -2,6-(C 6发现H 2 -2,4,6-i-Pr 3)2))位于单重态势能面的整体最小值上,并且在热力学上是稳定的。价电子键合模型表明,R'In
SbR'分子具有空间庞大的基团(R'= SiMe(Si t Bu 3)2,SiiPrDis 2,Tbt(= C 6 H 2 -2,4,6- {CH(SiMe 3)2 } 3)和Ar *(= C 6 H 3 -2,6-(C 6发现H 2 -2,4,6-i-Pr 3)2))位于单重态势能面的整体最小值上,并且在热力学上是稳定的。价电子键合模型表明,R'In ![[三重键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e002.gif) SbR'的键合性质可以表示为
SbR'的键合性质可以表示为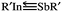 。我们基于几种理论方法(即电荷分解分析,自然键轨道分析和自然共振理论)的计算研究表明,较大取代基的电子和空间效应在制造三键R'In时都起着重要作用。
。我们基于几种理论方法(即电荷分解分析,自然键轨道分析和自然共振理论)的计算研究表明,较大取代基的电子和空间效应在制造三键R'In时都起着重要作用。![[三重键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e002.gif) SbR'物种可通过合成途径获得,并且可以稳定形式分离。
SbR'物种可通过合成途径获得,并且可以稳定形式分离。
更新日期:2018-03-19
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SbR molecules bearing suitable substituents (R). Calculations have demonstrated that the RIn
SbR molecules bearing suitable substituents (R). Calculations have demonstrated that the RIn![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SbR molecules possessing smaller substituents (such as R = F, OH, CH3, H, and SiH3) cannot be stabilized. Only the triple-bonded R′In
SbR molecules possessing smaller substituents (such as R = F, OH, CH3, H, and SiH3) cannot be stabilized. Only the triple-bonded R′In![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SbR′ molecules featuring sterically bulky groups (R′ = SiMe(SitBu3)2, SiiPrDis2, Tbt (= C6H2-2,4,6-{CH(SiMe3)2}3), and Ar* (= C6H3-2,6-(C6H2-2,4,6-i-Pr3)2)) are found to locate on the global minimum of the singlet potential energy surface and are thermodynamically stable. The valence-electron bonding model reveals that the bonding nature of R′In
SbR′ molecules featuring sterically bulky groups (R′ = SiMe(SitBu3)2, SiiPrDis2, Tbt (= C6H2-2,4,6-{CH(SiMe3)2}3), and Ar* (= C6H3-2,6-(C6H2-2,4,6-i-Pr3)2)) are found to locate on the global minimum of the singlet potential energy surface and are thermodynamically stable. The valence-electron bonding model reveals that the bonding nature of R′In![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SbR′ can be represented as
SbR′ can be represented as 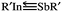 . Our computational investigations based on several theoretical methods (i.e., the charge decomposition analysis, the natural bond orbital analysis and the natural resonance theory) reveal that both the electronic and steric effects of bulkier substituent groups play important roles in making triple-bonded R′In
. Our computational investigations based on several theoretical methods (i.e., the charge decomposition analysis, the natural bond orbital analysis and the natural resonance theory) reveal that both the electronic and steric effects of bulkier substituent groups play important roles in making triple-bonded R′In![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) SbR′ species synthetically accessible and isolable in a stable form.
SbR′ species synthetically accessible and isolable in a stable form.
中文翻译:

可能的目标:
![[三重键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e002.gif) 具有高稳定性的三键合铟锑分子†
具有高稳定性的三键合铟锑分子†
我们已考虑开发
![[三重键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e002.gif) 带有合适取代基(R)的三键RIn SbR分子作为理论上的可能性。计算表明,
带有合适取代基(R)的三键RIn SbR分子作为理论上的可能性。计算表明,![[三重键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e002.gif) 具有较小取代基(如R = F,OH,CH 3,H和SiH 3)的RIn SbR分子无法稳定。只有三键结合的R'In
具有较小取代基(如R = F,OH,CH 3,H和SiH 3)的RIn SbR分子无法稳定。只有三键结合的R'In ![[三重键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e002.gif) SbR'分子具有空间庞大的基团(R'= SiMe(Si t Bu 3)2,SiiPrDis 2,Tbt(= C 6 H 2 -2,4,6- {CH(SiMe 3)2 } 3)和Ar *(= C 6 H 3 -2,6-(C 6发现H 2 -2,4,6-i-Pr 3)2))位于单重态势能面的整体最小值上,并且在热力学上是稳定的。价电子键合模型表明,R'In
SbR'分子具有空间庞大的基团(R'= SiMe(Si t Bu 3)2,SiiPrDis 2,Tbt(= C 6 H 2 -2,4,6- {CH(SiMe 3)2 } 3)和Ar *(= C 6 H 3 -2,6-(C 6发现H 2 -2,4,6-i-Pr 3)2))位于单重态势能面的整体最小值上,并且在热力学上是稳定的。价电子键合模型表明,R'In ![[三重键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e002.gif) SbR'的键合性质可以表示为
SbR'的键合性质可以表示为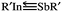 。我们基于几种理论方法(即电荷分解分析,自然键轨道分析和自然共振理论)的计算研究表明,较大取代基的电子和空间效应在制造三键R'In时都起着重要作用。
。我们基于几种理论方法(即电荷分解分析,自然键轨道分析和自然共振理论)的计算研究表明,较大取代基的电子和空间效应在制造三键R'In时都起着重要作用。![[三重键,长度为m-破折号]](https://www.rsc.org/images/entities/char_e002.gif) SbR'物种可通过合成途径获得,并且可以稳定形式分离。
SbR'物种可通过合成途径获得,并且可以稳定形式分离。











































 京公网安备 11010802027423号
京公网安备 11010802027423号