当前位置:
X-MOL 学术
›
Chem. Eur. J.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Rhodium(I)‐Catalyzed Ring‐Closing Reaction of Allene–Alkene–Alkynes: One‐Step Construction of Tricyclo[6.4.0.02,6] and Bicyclo[6.3.0] Skeletons from Linear Carbon Chains
Chemistry - A European Journal ( IF 3.9 ) Pub Date : 2018-04-16 , DOI: 10.1002/chem.201801239 Yasuaki Kawaguchi 1 , Asami Nagata 1 , Kei Kurokawa 1 , Haruna Yokosawa 1 , Chisato Mukai 1
Chemistry - A European Journal ( IF 3.9 ) Pub Date : 2018-04-16 , DOI: 10.1002/chem.201801239 Yasuaki Kawaguchi 1 , Asami Nagata 1 , Kei Kurokawa 1 , Haruna Yokosawa 1 , Chisato Mukai 1
Affiliation
|
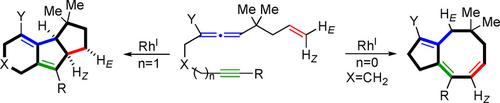
Treatment of dodecatrienyne derivatives with [RhCl(CO)2]2 in refluxing toluene effected the cycloisomerization to produce tricyclo[6.4.0.02,6]dodecadienes. The one‐carbon shortened undecatrienyne derivatives, however, afforded bicyclo[6.3.0]undecatriene derivatives instead of tricyclic compounds, the latter of which are well known as a basic skeleton of naturally occurring octanoids. On the basis of two experiments with deuterated substrates, a plausible reaction mechanism for the construction of these products was proposed.
中文翻译:

铑(I)催化的烯丙基-烯烃-炔烃的闭环反应:线性碳链上三环[6.4.0.02,6]和双环[6.3.0]骨架的一步构建
在回流的甲苯中用[RhCl(CO)2 ] 2处理十二碳三烯炔衍生物,实现了环异构化,生成了三环[6.4.0.0 2,6 ]十二碳二烯。然而,一碳短的十一碳三烯炔衍生物可以提供双环[6.3.0]十一碳三烯衍生物,而不是三环化合物,后者是众所周知的天然辛烷类化合物的基本骨架。基于氘化底物的两个实验,提出了构建这些产物的合理反应机理。
更新日期:2018-04-16
中文翻译:

铑(I)催化的烯丙基-烯烃-炔烃的闭环反应:线性碳链上三环[6.4.0.02,6]和双环[6.3.0]骨架的一步构建
在回流的甲苯中用[RhCl(CO)2 ] 2处理十二碳三烯炔衍生物,实现了环异构化,生成了三环[6.4.0.0 2,6 ]十二碳二烯。然而,一碳短的十一碳三烯炔衍生物可以提供双环[6.3.0]十一碳三烯衍生物,而不是三环化合物,后者是众所周知的天然辛烷类化合物的基本骨架。基于氘化底物的两个实验,提出了构建这些产物的合理反应机理。











































 京公网安备 11010802027423号
京公网安备 11010802027423号