Bioorganic Chemistry ( IF 5.1 ) Pub Date : 2018-01-31 , DOI: 10.1016/j.bioorg.2018.01.028 Mohamed A. Abdelgawad , Rania B. Bakr , Amany A. Azouz
|
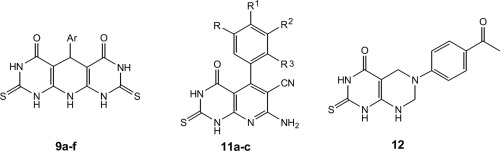
Some derivatives containing pyrido[2,3-d:6,5d′]dipyrimidine-4,5-diones (9a-f), tetrahydropyrido[2,3-d]pyrimidine-6-carbonitriles (11a-c) and 6-(4-acetylphenyl)-2-thioxo-2,3,5,6,7,8-hexahydro-1H-pyrimido[4,5-d]pyrimidin-4-one (12) were synthesized from 6-amino-2-thioxo-2,3-dihydro-1H-pyrimidin-4-one (8). The anti-inflammatory effect of these candidates was determined and the ulcer indices were calculated for active compounds. 7-Amino-5-(3,4,5-trimethoxyphenyl)-4-oxo-2-thioxo-1,2,3,4-tetrahydropyrido[2,3-d] pyrimidine-6-carbonitrile (11c) exhibited better edema inhibition than celecoxib. Moreover, compounds 9b, 9d and 11c revealed better COX-2 inhibitory activity in a range (IC50 = 0.25–0.89 µM) than celecoxib (IC50 = 1.11 µM). Regarding ulcerogenic liability, all of the compounds under the study were less ulcerogenic than indomethacin. Molecular docking studies had been carried on active candidates 9d and 11c to explore action mode of these candidates as leads for discovering other anti-inflammatory agents.
中文翻译:

新型嘧啶-吡啶杂化物:合成,环氧合酶抑制,抗炎活性和致溃疡性
一些含有吡啶并[2,3- d: 6,5 d' ]二嘧啶-4,5-二酮(9a-f),四氢吡啶并[2,3 - d ]嘧啶-6-腈(11a-c)和6的衍生物从6-氨基合成了-(4-乙酰苯基)-2-thioxo-2,3,5,6,7,8-六氢-1 H-嘧啶基[4,5- d ]嘧啶-4-酮(12) -2-硫代-2-3,3-二氢-1H-嘧啶-4-酮(8)。确定了这些候选药物的抗炎作用,并计算了活性化合物的溃疡指数。7-氨基-5-(3,4,5-三甲氧基苯基)-4-氧代-2-硫代-1,2,3,4-四氢吡啶并[2,3 - d ]嘧啶-6-腈(11c)显示出比塞来昔布更好的水肿抑制作用。此外,化合物9b,9d和11c在一定范围内(IC 50 = 0.25–0.89 µM)表现出比塞来昔布(IC 50 = 1.11 µM)更好的COX-2抑制活性。关于致溃疡的作用,本研究中所有化合物的致溃疡性均低于消炎痛。已经对活性候选物9d和11c进行了分子对接研究,以探索这些候选物的作用方式,作为发现其他抗炎剂的线索。



























 京公网安备 11010802027423号
京公网安备 11010802027423号