Dyes and Pigments ( IF 4.1 ) Pub Date : 2018-01-02 , DOI: 10.1016/j.dyepig.2017.12.065 Aloisio de A. Bartolomeu , Timothy J. Brocksom , Luiz C. da Silva Filho , Kleber T. de Oliveira
|
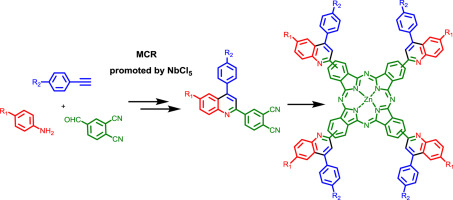
Herein, we demonstrate the efficiency of NbCl5 to promote a multicomponent reaction (MCR) for the synthesis of a library of phthalonitrile-quinoline dyads, which are very useful and new functionalized building blocks for phthalocyanine (PC) synthesis. Experimental mechanistic insights on the key MCR process are described, using a deuterated reagent, clearly showing the pericyclic nature of a hetero-Diels-Alder reaction. Examples of phthalocyanine (PC) syntheses were performed in order to demonstrate the versatility of the phthalonitrile-quinoline dyads. Preliminary photophysical measurements show that our phthalonitrile library is very promising for the production of new molecular scaffolds of PC derivatives with potential applications.
中文翻译:

NbCl 5介导的邻苯二甲腈-喹啉二元化合物的多组分反应:方法,范围,机理见解和在酞菁合成中的应用
在这里,我们证明了NbCl 5可以促进多组分反应(MCR)合成邻苯二甲腈-喹啉二元化合物的库,这对于酞菁(PC)合成是非常有用的和新的功能化构建基块。使用氘化试剂描述了关键MCR过程的实验机理,清楚地表明了杂Diels-Alder反应的周环性质。进行酞菁(PC)合成的示例,以证明邻苯二甲腈-喹啉二元化合物的多功能性。初步的光物理测量表明,我们的邻苯二甲腈文库对于具有潜在应用的PC衍生物新分子支架的生产非常有前途。











































 京公网安备 11010802027423号
京公网安备 11010802027423号