当前位置:
X-MOL 学术
›
J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Nickel-Catalyzed Reductive Etherification of Aldehydes at Room Temperature: C–O vs C–C Bond Formation
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2018-01-08 00:00:00 , DOI: 10.1021/acs.joc.7b02281 Sajjad Rahimi 1 , Farhad Panahi 1, 2 , Marzieh Bahmani 1 , Nasser Iranpoor 1
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2018-01-08 00:00:00 , DOI: 10.1021/acs.joc.7b02281 Sajjad Rahimi 1 , Farhad Panahi 1, 2 , Marzieh Bahmani 1 , Nasser Iranpoor 1
Affiliation
|
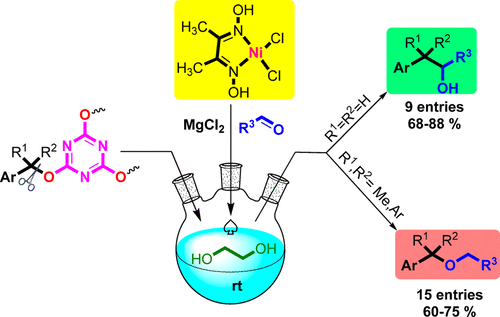
The reaction of secondary and tertiary benzyl alcohols activated by 2,4,6-trichloro-1,3,5-triazine (TCT) with aldehydes in the presence of NiCl2·dmg as a precatalyst in ethylene glycol afforded ethers at room temperature. A selective C–O vs C–C bond formation was observed for the secondary and tertiary benzyl alcohols in comparison with primary ones.
中文翻译:

镍在室温下催化醛的还原醚化:C–O与CC键的形成
在乙二醇中作为前催化剂的NiCl 2 ·dmg存在下,由2,4,6-三氯-1,3,5-三嗪(TCT)活化的仲和叔苄醇与醛的反应在室温下提供了醚。与伯醇相比,仲和叔苄醇具有选择性的C–O和C–C键形成。
更新日期:2018-01-08
中文翻译:

镍在室温下催化醛的还原醚化:C–O与CC键的形成
在乙二醇中作为前催化剂的NiCl 2 ·dmg存在下,由2,4,6-三氯-1,3,5-三嗪(TCT)活化的仲和叔苄醇与醛的反应在室温下提供了醚。与伯醇相比,仲和叔苄醇具有选择性的C–O和C–C键形成。











































 京公网安备 11010802027423号
京公网安备 11010802027423号