Tetrahedron ( IF 2.1 ) Pub Date : 2017-12-20 , DOI: 10.1016/j.tet.2017.12.039 Daria Nawrot , Marek Kolenič , Jiří Kuneš , Filip Kostelansky , Miroslav Miletin , Veronika Novakova , Petr Zimcik
|
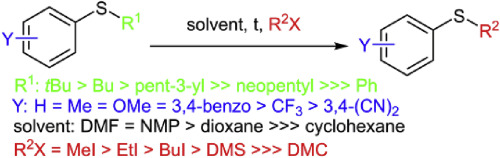
The reaction of methyl iodide with tert-butylphenylsulfide in DMF leads to a transalkylation that produces methylphenylsulfide. This transalkylation reaction was further studied by 1H NMR spectroscopy. The polarity of the solvent, the electron density on the sulfur atom, and the strength of the alkylating agent (MeI, EtI, BuI, dimethyl sulfate, or dimethyl carbonate) played important roles in the reaction. The suggested mechanism of the reaction involves the formation of a dialkyl aryl sulfonium salt that subsequently eliminates the radical. This mechanism was supported by the observation of higher conversion rates for compounds with more branched alkyl groups on the sulfur atom, which may lead to the formation of more stable radicals.
中文翻译:

烷基芳基硫醚与烷基化剂的烷基转移
甲基碘与叔丁基苯硫在DMF中的反应导致烷基转移,生成甲基苯硫。通过1 H NMR光谱进一步研究了该烷基转移反应。溶剂的极性,硫原子上的电子密度以及烷基化剂(MeI,EtI,BuI,硫酸二甲酯或碳酸二甲酯)的强度在反应中起着重要作用。所建议的反应机理涉及形成二烷基芳基formation盐,该盐随后消除了该基团。观察到硫原子上具有更多支链烷基的化合物具有更高的转化率,从而支持了该机理,这可能导致形成更稳定的自由基。



























 京公网安备 11010802027423号
京公网安备 11010802027423号