Bioorganic & Medicinal Chemistry Letters ( IF 2.5 ) Pub Date : 2017-12-20 , DOI: 10.1016/j.bmcl.2017.12.040 Anran Qian , Yazhou Zheng , Ruilian Wang , Jianhai Wei , Yongmei Cui , Xufeng Cao , Yushe Yang
|
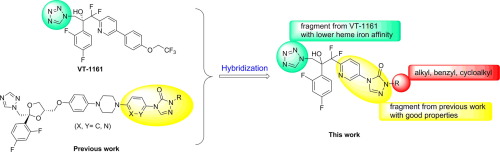
In this letter, we report our efforts to design, synthesize and evaluate biological activities of a series of novel hybridized compounds containing 1-tetrazole and 4-pyridinyl-1,2,4-triazole-3-one. An analysis of structure-activity data indicates that the target compounds with bulky and hydrophobic side chains exhibited stronger activities against the Candida spp and Cryptococcus neoformans tested than those of fluconazole and racemic VT-1161. Furthermore, 13k and 13ad were active against Microsporum gypseum, which was resistant to racemic VT-1161. In addition, 13k, 13ac and 13ad, with good in vitro activities against all of pathogenic fungi tested except for Aspergillus fumigatus, had no inhibition of human CYP3A4, suggesting a low risk of drug-drug interactions.
中文翻译:

具有强活性,宽广的抗真菌谱和高选择性的新型四唑类抗真菌剂的设计,合成及构效关系研究
在这封信中,我们报告了我们在设计,合成和评估一系列包含1-四唑和4-吡啶基-1,2,4-三唑-3-酮的新型杂交化合物的生物学活性方面所做的努力。对结构活性数据的分析表明,具有大分子和疏水侧链的目标化合物显示出对所测试的念珠菌和新隐隐球菌的活性比氟康唑和外消旋VT-1161的活性强。此外,13k和13ad对石膏微孢子菌具有活性,该石膏对消旋体VT-1161具有抗性。此外,13k,13ac和13ad具有良好的体外性能对除烟曲霉以外的所有致病真菌的活性没有抑制人CYP3A4的现象,提示药物与药物相互作用的风险较低。











































 京公网安备 11010802027423号
京公网安备 11010802027423号