Journal of Catalysis ( IF 7.3 ) Pub Date : 2017-12-14 , DOI: 10.1016/j.jcat.2017.11.004 Mark M. Sullivan , Aditya Bhan
|
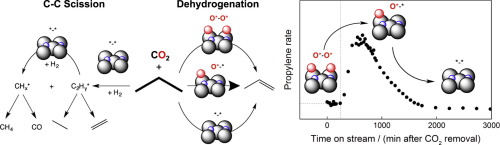
Mo2C catalyzes propane dehydrogenation and hydrogenolysis at 823 K; carbon selectivity can be tuned to >95% propylene via dehydrogenation in absence of H2, >95% CH4 via hydrogenolysis with H2 co-feed, or >80% CO via reforming pathways with H2 and CO2 co-feed. The changes in selectivity are mediated by an evolution in the coverage of oxidized (O∗) and carbidic (∗) surface sites which results in an evolution of O∗O∗, O∗
∗, and ∗
∗ site pairs that catalyze propane dehydrogenation. The fraction of O∗ in relation to ∗ was assessed from measured CO2/CO ratios because reverse water gas shift equilibrium exists under H2/CO2 co-feed steady state reaction conditions. Kinetic models based on the two-site dehydrogenation mechanism could be used to quantitatively describe measured rates of propane dehydrogenation at steady state with or without H2 and/or CO2 co-feed and the transient evolution in dehydrogenation rates upon removing H2 or CO2 in the influent stream to note that O∗
∗ site pairs exhibit the highest rate per gram. This model also provides a rationale for O∗ inhibition of H-activated hydrogenolysis pathways and for promotion of oxidative dehydrogenation rates with the introduction of hydrogen into CO2-propane influent streams. This study extends concepts developed for examining the catalytic effects of O∗ coverage on oxidative light alkane conversion from transition metal catalysts to also include carbidic formulations.
中文翻译:

氧覆盖率对碳化钼丙烷-CO 2反应速率和选择性的影响
Mo 2 C在823 K时催化丙烷脱氢和氢解。通过在不存在H 2的条件下进行脱氢,可以将碳的选择性提高至> 95%丙烯,通过与H 2共进料进行氢解,可以将碳选择性> 95%CH 4,或者可以通过与H 2和CO 2共进料的重整途径将> 80%的CO调整为丙烯。选择性的变化是由氧化(O ∗)和碳(∗)表面位点的覆盖范围的演变所介导的,这导致O ∗ O ∗,O ∗ ∗和∗ ∗位点对的发展,从而催化了丙烷的脱氢反应。O的分数*相对于*从测量CO评估2 / CO比,因为在H存在反向水煤气变换平衡2 / CO 2共进料稳态反应条件。基于两点脱氢机理的动力学模型可用于定量描述稳态条件下有或没有H 2和/或CO 2共进料时丙烷脱氢的测量速率,以及去除H 2或CO后脱氢速率的瞬时演变2进流要注意在其中O *
*网站对表现出每克的最高速率。该模型还提供了O ∗的基本原理通过将氢引入CO 2丙烷流入流中,抑制H激活的氢解途径并促进氧化脱氢速率。这项研究扩展了开发的概念,以研究O *覆盖率对过渡金属催化剂氧化轻烷烃转化的催化作用,还包括碳酸盐配方。



























 京公网安备 11010802027423号
京公网安备 11010802027423号