Catalysis Communications ( IF 3.4 ) Pub Date : 2017-12-02 , DOI: 10.1016/j.catcom.2017.11.014 Lina Jin , Xiaoshuang Zhao , Jian Ye , Xinye Qian , Mingdong Dong
|
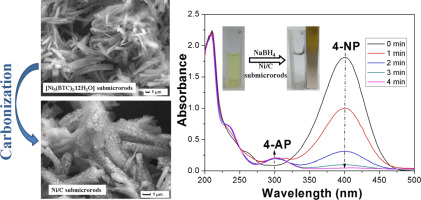
Magnetic Ni-carbon submicrorods were easily synthesized via the direct pyrolysis of [Ni3(BTC)2·12H2O] precursors at different temperatures. The [Ni3(BTC)2·12H2O] precursors were obtained using a simple direct precipitation method at room temperature, which is distinctly different from previous reports. The porous Ni-carbon submicrorods obtained exhibited excellent catalytic activity and good reusability for the reduction of 4-nitrophenol to 4-aminophenol. In particular, the activity factor κ (κ = k/m, s− 1 g− 1) of the Ni/C catalyst synthesized at 800 °C was found to be 17.41 s− 1 g− 1, which is higher than that of many previously reported Ni-based and noble metal-based catalysts. Therefore, magnetically recoverable Ni-carbon submicrorods appear as promising catalysts to be practically explored for the reduction of 4-nitrophenol because of their facile synthesis and excellent performance.
中文翻译:

MOF衍生的磁性Ni-碳亚微棒用于催化还原4-硝基苯酚
通过[Ni 3(BTC)2 ·12H 2 O]前体在不同温度下的直接热解,可以轻松合成磁性Ni-碳亚微棒。[Ni 3(BTC)2 ·12H 2 O]前体是使用简单的直接沉淀法在室温下获得的,这与以前的报道明显不同。获得的多孔Ni-碳亚微棒显示出优异的催化活性和良好的可重用性,可将4-硝基苯酚还原为4-氨基苯酚。特别地,发现在800℃下合成的Ni / C催化剂的活性因子κ(κ= k / m,s -1 g -1)为17.41 s -1 g -1,这比以前报道的许多镍基和贵金属基催化剂的要高。因此,可磁回收的Ni-碳亚微棒由于其合成容易且性能优异而成为有望用于还原4-硝基苯酚的有希望的催化剂。










































 京公网安备 11010802027423号
京公网安备 11010802027423号