Journal of Catalysis ( IF 6.5 ) Pub Date : 2017-12-09 , DOI: 10.1016/j.jcat.2017.11.027 J.P. Espinós , V.J. Rico , J. González-Cobos , J.R. Sánchez-Valencia , V. Pérez-Dieste , C. Escudero , A. de Lucas-Consuegra , A.R. González-Elipe
|
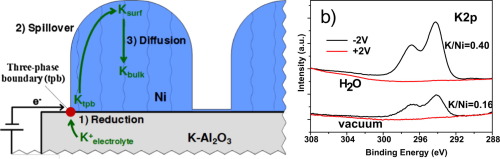
In this work we investigate by in-situ near-ambient pressure photoemission (NAPP) spectroscopy the phenomenon of Electrochemical Promotion of Catalysis (EPOC). We studied the reduction and diffusion kinetics of alkaline ions in a solid electrolyte cell formed by a nickel electrode supported on K+-β-alumina electrolyte. Experiments in ultra-high vacuum and in the presence of steam showed that the amount of potassium atoms supplied to the surface is probably affected by nickel electronic modifications induced by adsorbed OH− groups. It was also deduced that part of the segregated potassium would be adsorbed at inner interfaces where it would be inaccessible to the photoelectron analyzer. A migration mechanism of the promoter is proposed consisting in: (i) the electrochemical reduction of the alkali ions (potassium) at the Ni/solid electrolyte/gas interface; (ii) the spillover of potassium atoms onto the Ni gas-exposed surface; and (iii) the diffusion of potassium atoms to Ni inner grain boundary interfaces.
中文翻译:

原位监测电化学促进催化现象
在这项工作中,我们通过原位近环境压力光发射(NAPP)光谱研究了电化学促进催化作用(EPOC)的现象。我们研究了由K + -β-氧化铝电解质上支撑的镍电极形成的固体电解质电池中碱性离子的还原和扩散动力学。在超高真空和蒸汽的存在下的实验表明,钾原子供给到表面的量可能受到由吸附引起的OH镍电子修改-组。还推断出部分偏析的钾将被吸附在内部界面处,在该内部界面处光电子分析仪将无法接近。提出了促进剂的迁移机理,其包括:(i)Ni /固体电解质/气体界面处的碱金属离子(钾)的电化学还原;(ii)钾原子溢出到暴露于Ni气体的表面上;(iii)钾原子向Ni内部晶界界面的扩散。











































 京公网安备 11010802027423号
京公网安备 11010802027423号