当前位置:
X-MOL 学术
›
Org. Biomol. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
A Brønsted acid-promoted asymmetric intramolecular allylic amination of alcohols†
Organic & Biomolecular Chemistry ( IF 2.9 ) Pub Date : 2017-12-07 00:00:00 , DOI: 10.1039/c7ob02599h Jianqiao Zhou 1, 2, 3, 4, 5 , Hexin Xie 1, 2, 3, 4, 5
Organic & Biomolecular Chemistry ( IF 2.9 ) Pub Date : 2017-12-07 00:00:00 , DOI: 10.1039/c7ob02599h Jianqiao Zhou 1, 2, 3, 4, 5 , Hexin Xie 1, 2, 3, 4, 5
Affiliation
|
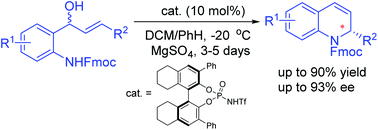
Reported herein is a chiral Brønsted acid-catalyzed asymmetric intramolecular allylic amination reaction, allowing facile access to a range of biologically interesting chiral 2-substituted hydroquinolines in up to 90% yield and with up to 93% ee. Furthermore, a significant effect of an N-protecting group was observed in this asymmetric process.
中文翻译:

布朗斯台德酸促进的醇不对称分子内烯丙基胺化†
本文报道的是手性布朗斯台德酸催化的不对称分子内烯丙基胺化胺化反应,可轻松获得高达90%的收率和93%ee的一系列生物学上有趣的手性2-取代氢喹啉。此外,在该不对称过程中观察到了N-保护基的显着作用。
更新日期:2017-12-07
中文翻译:

布朗斯台德酸促进的醇不对称分子内烯丙基胺化†
本文报道的是手性布朗斯台德酸催化的不对称分子内烯丙基胺化胺化反应,可轻松获得高达90%的收率和93%ee的一系列生物学上有趣的手性2-取代氢喹啉。此外,在该不对称过程中观察到了N-保护基的显着作用。











































 京公网安备 11010802027423号
京公网安备 11010802027423号