当前位置:
X-MOL 学术
›
J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Diastereoselective Synthesis of Functionalized Diketopiperazines through Post-transformational Reactions
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2017-11-07 00:00:00 , DOI: 10.1021/acs.joc.7b01855 Saeed Balalaie 1, 2 , Reihaneh Ramezani Kejani 1 , Elmira Ghabraie 1 , Fatemeh Darvish 1 , Frank Rominger 3 , Fatima Hamdan 1 , Hamid Reza Bijanzadeh 4
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2017-11-07 00:00:00 , DOI: 10.1021/acs.joc.7b01855 Saeed Balalaie 1, 2 , Reihaneh Ramezani Kejani 1 , Elmira Ghabraie 1 , Fatemeh Darvish 1 , Frank Rominger 3 , Fatima Hamdan 1 , Hamid Reza Bijanzadeh 4
Affiliation
|
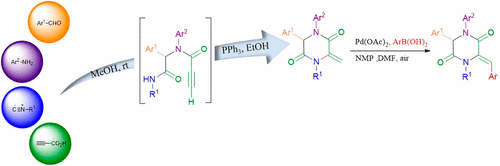
A diversity-oriented access to diastereoselective arylidene 2,5-diketopiperazines is elaborated via a sequential Ugi post-transformation involving catalytic cyclization and oxidative Heck reaction sequence. This sequence offers an interesting multicomponent entry to a library of 2,5-diketopiperazines and arylidene 2,5-diketopiperazines under mild reaction conditions in good to excellent yields.
中文翻译:

通过转化后反应进行非对映选择性合成功能化二酮哌嗪
通过涉及催化环化和氧化性Heck反应序列的顺序Ugi转化,详细介绍了非对映选择性亚芳基2,5-二酮哌嗪的多样性导向。在温和的反应条件下,该序列为2,5-二酮哌嗪和亚芳基2,5-二酮哌嗪的文库提供了有趣的多组分输入,收率很高,而且收率极好。
更新日期:2017-11-08
中文翻译:

通过转化后反应进行非对映选择性合成功能化二酮哌嗪
通过涉及催化环化和氧化性Heck反应序列的顺序Ugi转化,详细介绍了非对映选择性亚芳基2,5-二酮哌嗪的多样性导向。在温和的反应条件下,该序列为2,5-二酮哌嗪和亚芳基2,5-二酮哌嗪的文库提供了有趣的多组分输入,收率很高,而且收率极好。











































 京公网安备 11010802027423号
京公网安备 11010802027423号