当前位置:
X-MOL 学术
›
J. Org. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Chemoselective α-Methylenation of Aromatic Ketones Using the NaAuCl4/Selectfluor/DMSO System
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2017-11-07 00:00:00 , DOI: 10.1021/acs.joc.7b01790 Hongbo Zhu 1 , Xin Meng 1 , Yanhui Zhang 1 , Guang Chen 1, 2 , Ziping Cao 1, 2 , Xuejun Sun 1, 2 , Jinmao You 1, 2
The Journal of Organic Chemistry ( IF 3.3 ) Pub Date : 2017-11-07 00:00:00 , DOI: 10.1021/acs.joc.7b01790 Hongbo Zhu 1 , Xin Meng 1 , Yanhui Zhang 1 , Guang Chen 1, 2 , Ziping Cao 1, 2 , Xuejun Sun 1, 2 , Jinmao You 1, 2
Affiliation
|
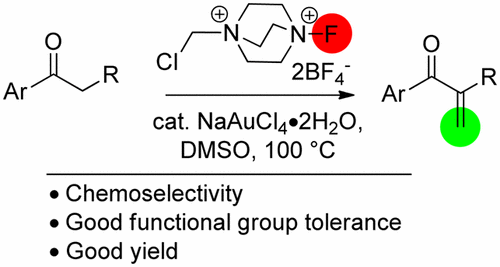
Gold-catalyzed chemoselective α-methylenation of aromatic ketones was developed through the use of Selectfluor as a methylenating agent. A variety of useful 1,2-disubstituted propenone derivatives can be prepared in good yields via the present protocol. This reaction features a simple operation, good functional group tolerance, and broad scope of substrates.
中文翻译:

NaAuCl 4 / Selectfluor / DMSO系统对芳族酮的化学选择性α-甲基化
通过使用Selectfluor作为亚甲基化剂,开发了金催化的芳香族酮的化学选择性α-甲基化反应。通过本方案,可以以高收率制备各种有用的1,2-二取代的丙烯酮衍生物。该反应具有操作简单,官能团耐受性好和底物范围广的特点。
更新日期:2017-11-07
中文翻译:

NaAuCl 4 / Selectfluor / DMSO系统对芳族酮的化学选择性α-甲基化
通过使用Selectfluor作为亚甲基化剂,开发了金催化的芳香族酮的化学选择性α-甲基化反应。通过本方案,可以以高收率制备各种有用的1,2-二取代的丙烯酮衍生物。该反应具有操作简单,官能团耐受性好和底物范围广的特点。











































 京公网安备 11010802027423号
京公网安备 11010802027423号