Biomaterials ( IF 12.8 ) Pub Date : 2017-10-27 , DOI: 10.1016/j.biomaterials.2017.10.039 Yang Wang , Sheng Yin , Li Zhang , Kairong Shi , Jiajing Tang , Zhirong Zhang , Qin He
|
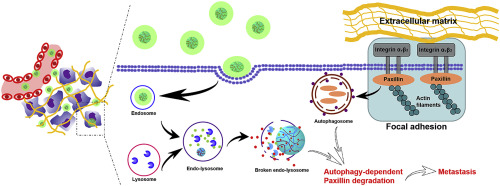
In attempts to explore the role of autophagy in breast cancer metastasis, we here report a tumor-activatable particle (named as “D/PSP@CQ/CaP”) with the ability of efficient autophagy inhibition. D/PSP@CQ/CaP was prepared by coprecipitating chloroquine phosphate (CQ) with calcium chloride, in the form of chloroquine-calcium phosphate coprecipitate (CQ/CaP), onto the surface of a deep-tumor-penetrating doxorubicine (DOX)-loading core particle (named as “D/PSP”). CQ/CaP could partly disintegrate and release CQ within tumor microenvironment and totally be dissolved within lysosomes. Paxillin is a key component of focal adhesion which functions to anchor tumors cells within the primary tumor for limiting cancer cells' detachment from the primary tumor. We tested that autophagy inhibition caused by CQ released from CQ/CaP could reduce the degradation of paxillin by 2.9 folds in vitro and 2.5 folds in vivo (vs. Control), respectively. Thus metastasis could be influenced by exploiting autophagy-dependent paxillin degradation. Data analysis together proved that D/PSP@CQ/CaP decreased the cancer metastatic extent by 7.5 folds (vs. Control) on mice model via inhibiting the autophagy-dependent disassembly of focal adhesion. At the same time, the growth rate of tumors treated by D/PSP@CQ/CaP was inhibited by 9.1 folds (vs. Control), which could be attributed to its effective tumor drug delivery.
中文翻译:

通过抑制自噬依赖性黏着斑的分解,可在乳腺癌中产生具有抗转移潜力的可激活肿瘤的颗粒
为了探索自噬在乳腺癌转移中的作用,我们在此报告了一种具有有效自噬抑制能力的肿瘤可激活颗粒(称为“ D / PSP @ CQ / CaP”)。D / PSP @ CQ / CaP的制备方法是:将氯喹磷酸酯(CQ)与氯化钙以氯喹-磷酸钙共沉淀物(CQ / CaP)的形式共沉淀到深肿瘤穿透性阿霉素(DOX)-的表面上。加载核心粒子(命名为“ D / PSP”)。CQ / CaP可以在肿瘤微环境中部分分解并释放CQ,并完全溶解在溶酶体内。Paxillin是粘着斑的关键组成部分,其作用是将肿瘤细胞锚定在原发性肿瘤内,以限制癌细胞与原发性肿瘤的分离。分别是体外和体内的2.5倍(相对于对照)。因此,转移可以通过利用自噬依赖性的Paxillin降解来影响。数据分析共同证明,D / PSP @ CQ / CaP通过抑制自噬依赖性黏着斑的分解,使小鼠模型的癌症转移程度降低了7.5倍(相对于对照)。同时,用D / PSP @ CQ / CaP处理的肿瘤的生长速率被抑制了9.1倍(相对于对照),这可以归因于其有效的肿瘤药物递送。











































 京公网安备 11010802027423号
京公网安备 11010802027423号