当前位置:
X-MOL 学术
›
Org. Chem. Front.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Rhodium(III)-catalyzed synthesis of indanones via C–H activation of phenacyl phosphoniums and coupling with olefins
Organic Chemistry Frontiers ( IF 4.6 ) Pub Date : 2017-07-20 00:00:00 , DOI: 10.1039/c7qo00510e Yunyun Li 1, 2, 3, 4, 5 , Xifa Yang 1, 2, 3, 4, 5 , Lingheng Kong 1, 2, 3, 4, 5 , Xingwei Li 1, 2, 3, 4
Organic Chemistry Frontiers ( IF 4.6 ) Pub Date : 2017-07-20 00:00:00 , DOI: 10.1039/c7qo00510e Yunyun Li 1, 2, 3, 4, 5 , Xifa Yang 1, 2, 3, 4, 5 , Lingheng Kong 1, 2, 3, 4, 5 , Xingwei Li 1, 2, 3, 4
Affiliation
|
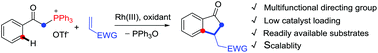
Phosphonium ylide acts as an efficient bifunctional directing group in Rh(III)-catalyzed C–H activation of arenes and oxidative coupling with activated olefins, leading to facile construction of indanones via a sequence of oxidative olefination and carboannulation. The phosphonium moiety functions as an oxophilic group, and dephosphination triggers a nucleophilic cyclization.
中文翻译:

铑(III)茚满酮的合成催化的经由苯甲酰甲基鏻的C-H活化和与烯烃偶合
内鎓盐膦在Rh(III)催化的芳烃的C–H活化和与活化烯烃的氧化偶联中充当有效的双官能团导向基团,从而通过一系列氧化烯化和碳环化反应而轻松地构建茚满酮。moiety部分起亲氧基团的作用,并且脱磷触发亲核环化。
更新日期:2017-10-25
中文翻译:

铑(III)茚满酮的合成催化的经由苯甲酰甲基鏻的C-H活化和与烯烃偶合
内鎓盐膦在Rh(III)催化的芳烃的C–H活化和与活化烯烃的氧化偶联中充当有效的双官能团导向基团,从而通过一系列氧化烯化和碳环化反应而轻松地构建茚满酮。moiety部分起亲氧基团的作用,并且脱磷触发亲核环化。











































 京公网安备 11010802027423号
京公网安备 11010802027423号