当前位置:
X-MOL 学术
›
Adv. Synth. Catal.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
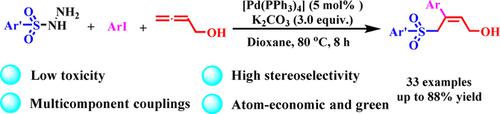
Palladium‐Catalyzed Three‐Component Tandem Reaction for One‐pot Highly Stereoselective Synthesis of (Z)‐α‐Hydroxymethyl Allylic Sulfones
Advanced Synthesis & Catalysis ( IF 4.4 ) Pub Date : 2017-11-06 , DOI: 10.1002/adsc.201701011 Yunlei Hou 1 , Qi Shen 1 , Zefei Li 1 , Shaowei Chen 1 , Yanfang Zhao 1 , Mingze Qin 1 , Ping Gong 1
Advanced Synthesis & Catalysis ( IF 4.4 ) Pub Date : 2017-11-06 , DOI: 10.1002/adsc.201701011 Yunlei Hou 1 , Qi Shen 1 , Zefei Li 1 , Shaowei Chen 1 , Yanfang Zhao 1 , Mingze Qin 1 , Ping Gong 1
Affiliation
|
A novel method is reported for the stereoselective synthesis of highly functionalized allyl aryl sulfones. This protocol is based on a Pd‐catalyzed three‐component tandem reaction of sulfonyl hydrazides and aryl iodides with allenes and exhibits high (Z)‐selectivity, good yields, minimal waste, ample product scope, and operational simplicity.
中文翻译:

一锅高立体选择性合成(Z)-α-羟甲基烯丙基砜的钯催化三组分串联反应
报道了一种新方法用于立体选择性合成高度官能化的烯丙基芳基砜。该方案基于磺酰肼和芳基碘化物与丙二烯的Pd催化三组分串联反应,并显示出高(Z)-选择性,良好的收率,最小的浪费,丰富的产品范围和操作简便性。
更新日期:2017-11-06
中文翻译:

一锅高立体选择性合成(Z)-α-羟甲基烯丙基砜的钯催化三组分串联反应
报道了一种新方法用于立体选择性合成高度官能化的烯丙基芳基砜。该方案基于磺酰肼和芳基碘化物与丙二烯的Pd催化三组分串联反应,并显示出高(Z)-选择性,良好的收率,最小的浪费,丰富的产品范围和操作简便性。











































 京公网安备 11010802027423号
京公网安备 11010802027423号