Journal of Fluorine Chemistry ( IF 1.7 ) Pub Date : 2017-10-20 , DOI: 10.1016/j.jfluchem.2017.10.006 Zita Tokárová , Róbert Balogh , Pavol Tisovský , Katarína Hrnčariková , Daniel Végh
|
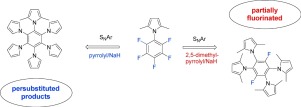
A series of novel N-pyrrolyl-, N-(2,5-dimethyl)pyrrolyl- and N-(2,5-dithiophen-2-yl)pyrrolyl- partially fluorinated and/or fully substituted benzenes have been prepared by the nucleophilic substitution of fluorine (SNAr) in hexafluorobenzene, 1-pentafluorophenyl-1H-pyrrole; 2,5-dimethyl-1-pentafluorophenyl-1H-pyrrole; 2,5-di(thiophen-2-yl)pentafluorophenyl and 2,3,5,6-tetrafluoroterephtalonitrile. The observed substitution pattern indicates the gradual defluorination of particular fluorine atoms depending on the nature and amount of the used nucleophile (pyrrole/NaH vs. 2,5-dimethylpyrrole/NaH).
中文翻译:

用吡咯和2,5-二甲基吡咯直接亲核取代多氟苯
通过亲核反应制备了一系列新颖的N-吡咯基- ,N-(2,5-二甲基)吡咯基-和N-(2,5-二噻吩-2-基)吡咯基-部分氟化和/或完全取代的苯。氟取代(S ñ在六氟苯的Ar),1-五氟苯基-1H-吡咯; 2,5-二甲基-1-五氟苯基-1H-吡咯; 2,5-二(噻吩-2-基)五氟苯基和2,3,5,6-四氟对苯二甲腈。观察到的取代模式表明特定氟原子逐渐脱氟,具体取决于所用亲核试剂的性质和数量(吡咯/ NaH与2,5-二甲基吡咯/ NaH)。











































 京公网安备 11010802027423号
京公网安备 11010802027423号