当前位置:
X-MOL 学术
›
Org. Lett.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Total Synthesis of (−)-Geissoschizol through Ir-Catalyzed Allylic Amidation as the Key Step
Organic Letters ( IF 4.9 ) Pub Date : 2017-11-02 00:00:00 , DOI: 10.1021/acs.orglett.7b03133 Yu Zheng 1, 2 , Kun Wei 1 , Yu-Rong Yang 1
Organic Letters ( IF 4.9 ) Pub Date : 2017-11-02 00:00:00 , DOI: 10.1021/acs.orglett.7b03133 Yu Zheng 1, 2 , Kun Wei 1 , Yu-Rong Yang 1
Affiliation
|
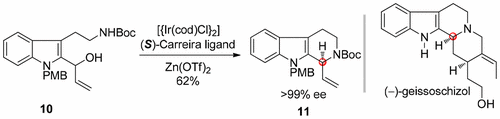
The iridium-catalyzed, Zn(OTf)2 promoted asymmetric allylic amidation of a secondary alcohol derived from tryptamine has been utilized to forge a tetrahydro-β-carboline. Based on this key step, a novel, facile, and enantioselective total synthesis of (−)-geissoschizol was achieved in 10 steps.
中文翻译:

以Ir催化的烯丙基酰胺化为关键的(-)-Geissoschizol的全合成。
铱催化的Zn(OTf)2促进的不对称烯丙基酰胺化衍生自色胺的仲醇已被用于伪造四氢-β-咔啉。基于该关键步骤,分十步完成了新颖,简便,对映选择性的(-)-geissoschizol合成。
更新日期:2017-11-02
中文翻译:

以Ir催化的烯丙基酰胺化为关键的(-)-Geissoschizol的全合成。
铱催化的Zn(OTf)2促进的不对称烯丙基酰胺化衍生自色胺的仲醇已被用于伪造四氢-β-咔啉。基于该关键步骤,分十步完成了新颖,简便,对映选择性的(-)-geissoschizol合成。











































 京公网安备 11010802027423号
京公网安备 11010802027423号