当前位置:
X-MOL 学术
›
Org. Lett.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Co-Catalyzed C(sp3)–H Oxidative Coupling of Glycine and Peptide Derivatives
Organic Letters ( IF 4.9 ) Pub Date : 2017-09-22 00:00:00 , DOI: 10.1021/acs.orglett.7b02567 Marcos San Segundo 1 , Itziar Guerrero 1 , Arkaitz Correa 1
Organic Letters ( IF 4.9 ) Pub Date : 2017-09-22 00:00:00 , DOI: 10.1021/acs.orglett.7b02567 Marcos San Segundo 1 , Itziar Guerrero 1 , Arkaitz Correa 1
Affiliation
|
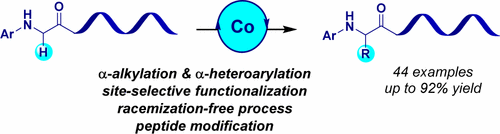
Cobalt-catalyzed selective α-alkylation and α-heteroarylation processes of α-amino esters and peptide derivatives are described. These cross-dehydrogenative reactions occur under mild conditions and allow for the rapid assembly of structurally diverse α-amino carbonyl compounds. Unlike enolate chemistry, these methods are distinguished by their site-specificity, occur without racemization of the existing chiral centers, and exhibit total selectivity for aryl glycine motifs over other amino acid units, hence providing ample opportunities for peptide modifications.
中文翻译:

甘氨酸和肽衍生物的共催化C(sp 3)–H氧化偶联
描述了钴催化的α-氨基酯和肽衍生物的选择性α-烷基化和α-杂芳基化过程。这些交叉脱氢反应在温和的条件下发生,并允许结构多样的α-氨基羰基化合物的快速组装。与烯醇式化学不同,这些方法的特征在于其位点特异性,在不消旋现有手性中心的情况下发生,并且相对于其他氨基酸单元表现出对芳基甘氨酸基序的总选择性,因此为肽修饰提供了充足的机会。
更新日期:2017-09-22
中文翻译:

甘氨酸和肽衍生物的共催化C(sp 3)–H氧化偶联
描述了钴催化的α-氨基酯和肽衍生物的选择性α-烷基化和α-杂芳基化过程。这些交叉脱氢反应在温和的条件下发生,并允许结构多样的α-氨基羰基化合物的快速组装。与烯醇式化学不同,这些方法的特征在于其位点特异性,在不消旋现有手性中心的情况下发生,并且相对于其他氨基酸单元表现出对芳基甘氨酸基序的总选择性,因此为肽修饰提供了充足的机会。











































 京公网安备 11010802027423号
京公网安备 11010802027423号