当前位置:
X-MOL 学术
›
Tetrahedron
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Synthesis of the AE bicyclic of Daphniphyllum alkaloid yuzurine
Tetrahedron ( IF 2.1 ) Pub Date : 2017-09-20 , DOI: 10.1016/j.tet.2017.09.035 Yue Meng Liu , Fei Li , Qi Wang , Jun Yang
中文翻译:

的AE双环的合成虎皮楠生物碱yuzurine
更新日期:2017-09-20
Tetrahedron ( IF 2.1 ) Pub Date : 2017-09-20 , DOI: 10.1016/j.tet.2017.09.035 Yue Meng Liu , Fei Li , Qi Wang , Jun Yang
|
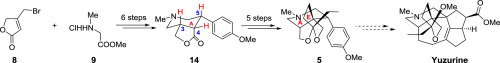
A concise synthesis of the AE bicyclic system of Daphniphyllum alkaloid yuzurine is described. The synthetic sequence features a Dieckmann condensation to construct ring A, reduction of α, β-unsaturated double bond and propargylation of α-position of lactone to diastereoselectively generate relative stereocenter C-3, C-4 and C-5, and an acid-catalyzed cyclization to assemble ring E.
中文翻译:

的AE双环的合成虎皮楠生物碱yuzurine
的AE双环系统的简明合成虎皮生物碱yuzurine进行说明。合成序列的特征是通过Dieckmann缩合反应来构建环A,还原α,β-不饱和双键和内酯的α-位炔丙基化以非对映选择性地产生相对的立体中心C-3,C-4和C-5,以及酸-催化环化以组装环E。











































 京公网安备 11010802027423号
京公网安备 11010802027423号