当前位置:
X-MOL 学术
›
Adv. Synth. Catal.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Organocatalytic Asymmetric Synthesis of 3,3-Disubstituted 3,4-Dihydro-2-quinolones
Advanced Synthesis & Catalysis ( IF 4.4 ) Pub Date : 2017-09-20 06:06:24 , DOI: 10.1002/adsc.201700655 Soumendranath Mukhopadhyay 1 , Utpal Nath 1 , Subhas Chandra Pan 1
Advanced Synthesis & Catalysis ( IF 4.4 ) Pub Date : 2017-09-20 06:06:24 , DOI: 10.1002/adsc.201700655 Soumendranath Mukhopadhyay 1 , Utpal Nath 1 , Subhas Chandra Pan 1
Affiliation
|
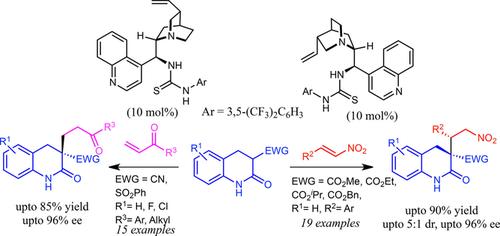
The first organocatalytic asymmetric reaction employing 3,4-dihydro-2-quinolone has been developed leading to the synthesis of biologically important 3,3-disubstituted-dihydro-2-quinolones. Cinchona alkaloid derived bifunctional amino-thiourea catalysts were found to be the best catalysts. The products were obtained in high enantio- and good diastereoselectivities and also few synthetic transformations have been demonstrated.
中文翻译:

3,3-二取代的3,4-二氢-2-喹诺酮类化合物的有机催化不对称合成
已经开发了使用3,4-二氢-2-喹诺酮的第一有机催化不对称反应,从而导致了生物学上重要的3,3-二取代-二氢-2-喹诺酮的合成。发现金鸡纳生物碱衍生的双官能氨基-硫脲催化剂是最好的催化剂。得到的产物具有高对映体和良好的非对映选择性,也已证明了很少的合成转化。
更新日期:2017-09-20
中文翻译:

3,3-二取代的3,4-二氢-2-喹诺酮类化合物的有机催化不对称合成
已经开发了使用3,4-二氢-2-喹诺酮的第一有机催化不对称反应,从而导致了生物学上重要的3,3-二取代-二氢-2-喹诺酮的合成。发现金鸡纳生物碱衍生的双官能氨基-硫脲催化剂是最好的催化剂。得到的产物具有高对映体和良好的非对映选择性,也已证明了很少的合成转化。











































 京公网安备 11010802027423号
京公网安备 11010802027423号