当前位置:
X-MOL 学术
›
Org. Lett.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Sonogashira Reaction Using Arylsulfonium Salts as Cross-Coupling Partners
Organic Letters ( IF 5.2 ) Pub Date : 2017-09-20 00:00:00 , DOI: 10.1021/acs.orglett.7b02764 Ze-Yu Tian 1 , Shi-Meng Wang 1 , Su-Jiao Jia 1 , Hai-Xia Song 1 , Cheng-Pan Zhang 1
Organic Letters ( IF 5.2 ) Pub Date : 2017-09-20 00:00:00 , DOI: 10.1021/acs.orglett.7b02764 Ze-Yu Tian 1 , Shi-Meng Wang 1 , Su-Jiao Jia 1 , Hai-Xia Song 1 , Cheng-Pan Zhang 1
Affiliation
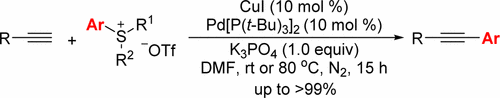
|
Triarylsulfonium, alkyl- and fluoroalkyl(diaryl)sulfonium, and aryl(dialkyl)sulfonium triflates are successfully used as a new family of cross-coupling participants in the Sonogashira reaction as aryldiazonium, diaryliodonium, and tetraphenylphosphonium salts. It was found that terminal alkynes reacted mildly with triarylsulfonium or (2,2,2-trifluoroethyl)diphenylsulfonium triflate at room temperature under Pd- and Cu-cocatalysis to give the corresponding arylalkynes in up to >99% yield. This protocol represents the first use of arylsulfonium salts as cross-coupling partners in the Pd/Cu-catalyzed Sonogashira reaction.
中文翻译:

使用芳基ulf盐作为交叉偶联伙伴的Sonogashira反应
三芳基ulf,烷基和氟烷基(二芳基)ulf和芳基(二烷基)ulf三氟甲磺酸盐已成功用作Sonogashira反应中的交叉偶联新家族,如芳基重氮盐,二芳基碘鎓盐和四苯基phosph盐。发现末端炔烃在室温下在Pd-和Cu-共催化下与三芳基(或三氟甲磺酸(2,2,2-三氟乙基)二苯基mild轻度反应,以高达> 99%的产率得到相应的芳基炔。该方案代表在Pd / Cu催化的Sonogashira反应中首次使用芳基ulf盐作为交叉偶联伴侣。
更新日期:2017-09-20
中文翻译:

使用芳基ulf盐作为交叉偶联伙伴的Sonogashira反应
三芳基ulf,烷基和氟烷基(二芳基)ulf和芳基(二烷基)ulf三氟甲磺酸盐已成功用作Sonogashira反应中的交叉偶联新家族,如芳基重氮盐,二芳基碘鎓盐和四苯基phosph盐。发现末端炔烃在室温下在Pd-和Cu-共催化下与三芳基(或三氟甲磺酸(2,2,2-三氟乙基)二苯基mild轻度反应,以高达> 99%的产率得到相应的芳基炔。该方案代表在Pd / Cu催化的Sonogashira反应中首次使用芳基ulf盐作为交叉偶联伴侣。



























 京公网安备 11010802027423号
京公网安备 11010802027423号