Bioorganic & Medicinal Chemistry ( IF 3.3 ) Pub Date : 2017-09-05 , DOI: 10.1016/j.bmc.2017.08.049 Sourabh Mundra , Vandana Thakur , Angelica M. Bello , Sumit Rathore , Mohd Asad , Lianhu Wei , Jane Yang , Sai Kumar Chakka , Radhakrishnan Mahesh , Pawan Malhotra , Asif Mohmmed , Lakshmi P. Kotra
|
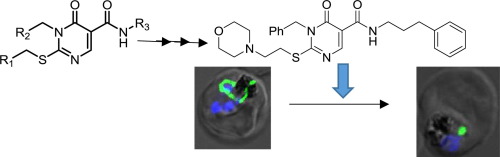
The prokaryotic ATP-dependent ClpP protease, localized in the relict plastid of malaria parasite, represents a potential drug target. In the present study, we utilized in silico structure-based screening and medicinal chemistry approaches to identify a novel pyrimidine series of compounds inhibiting P. falciparum ClpP protease activity and evaluated their antiparasitic activities. Structure-activity relationship indicated that morpholine moiety at C2, an aromatic substitution at N3 and a 4-oxo moiety on the pyrimidine are important for potent inhibition of ClpP enzyme along with antiparasiticidal activity. Compound 33 exhibited potent antiparasitic activity (EC₅₀ 9.0 ± 0.2 μM), a 9-fold improvement over the antiparasitic activity of the hit molecule 6. Treatment of blood stage P. falciparum cultures with compound 33 caused morphological and developmental abnormalities in the parasites; further, compound 33 treatment hindered apicoplast development indicating the targeting of apicoplast.
中文翻译:

一类新的疟原虫CLPP蛋白酶抑制剂作为潜在抗疟药
定位于疟原虫的残质体中的原核ATP依赖性ClpP蛋白酶代表了潜在的药物靶标。在本研究中,我们利用基于计算机结构的筛选和药物化学方法,鉴定了一种新型嘧啶系列化合物,可抑制恶性疟原虫ClpP蛋白酶的活性并评估其抗寄生虫活性。构效关系表明,C2上的吗啉部分,N3上的芳香取代基和嘧啶上的4-氧代部分对于有效抑制ClpP酶及抗寄生虫活性很重要。化合物33表现出强效的抗寄生虫活性(EC₅₀9.0±0.2μM),比命中分子的抗寄生虫活性提高了9倍6。用化合物33处理恶性疟原虫血液培养物会引起寄生虫的形态和发育异常;此外,化合物33的治疗阻碍了蜂胶体的发育,表明了对蜂胶体的靶向。











































 京公网安备 11010802027423号
京公网安备 11010802027423号