当前位置:
X-MOL 学术
›
Tetrahedron
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
Dual-reagent organophosphine catalyzed asymmetric Mannich reactions of isocyanoacetates with N-Boc-aldimines
Tetrahedron ( IF 2.1 ) Pub Date : 2017-08-30 , DOI: 10.1016/j.tet.2017.08.031 Xiong Ji , Wei-Guo Cao , Gang Zhao
中文翻译:

双试剂有机膦催化异氰酸酯与N -Boc-醛亚胺的不对称曼尼希反应
更新日期:2017-08-30
Tetrahedron ( IF 2.1 ) Pub Date : 2017-08-30 , DOI: 10.1016/j.tet.2017.08.031 Xiong Ji , Wei-Guo Cao , Gang Zhao
|
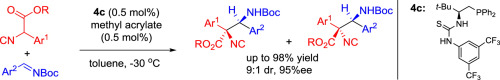
A combination of an amino-acid derived chiral phosphine catalyst and methyl acrylate has been employed to catalyze the direct Mannich reaction of α-aryl isocyanoacetate and N-Boc-aldimines efficiently. The loading of the catalyst could be as low as 0.5 mol% without compromise on the yield and enantioselectivity and the corresponding chiral adducts were obtained in excellent yields (up to 98%) and good enantioselectivities (up to 95%).
中文翻译:

双试剂有机膦催化异氰酸酯与N -Boc-醛亚胺的不对称曼尼希反应
氨基酸衍生的手性膦催化剂和丙烯酸甲酯的组合已被用来有效地催化α-芳基异氰基乙酸酯和N -Boc-醛亚胺的直接曼尼希反应。催化剂的负载量可以低至0.5mol%,而不会影响收率和对映选择性,并且以优异的收率(最高98%)和良好的对映选择性(最高95%)获得了相应的手性加合物。











































 京公网安备 11010802027423号
京公网安备 11010802027423号