当前位置:
X-MOL 学术
›
Chem. Commun.
›
论文详情
Our official English website, www.x-mol.net, welcomes your
feedback! (Note: you will need to create a separate account there.)
A cascade Claisen rearrangement/o-quinone methide formation/electrocyclization approach to 2H-chromenes
Chemical Communications ( IF 4.3 ) Pub Date : 2017-05-15 00:00:00 , DOI: 10.1039/c7cc03037a Liyan Song,Fang Huang,Liwen Guo,Ming-An Ouyang,Rongbiao Tong
Chemical Communications ( IF 4.3 ) Pub Date : 2017-05-15 00:00:00 , DOI: 10.1039/c7cc03037a Liyan Song,Fang Huang,Liwen Guo,Ming-An Ouyang,Rongbiao Tong
|
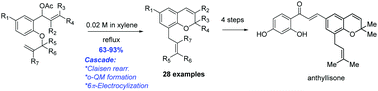
A new approach has been developed for the synthesis of 8-substituted 2H-chromenes, featuring a novel cascade aromatic Claisen rearrangement/o-quinone methide formation/6π-electrocyclization. This new method was demonstrated with 28 examples tolerating different substitutions at alkenes, allylic and aromatic ring and with total syntheses of three 2H-chromene natural products.
中文翻译:

级联Claisen重排/ ö -quinone甲基化物形成/ electrocyclization方法2个ħ -chromenes
已经开发出用于合成8-取代的2 H-色烯的新方法,其特征在于新颖的级联芳族克莱森重排/邻醌甲基化物形成/6π-电环化。28个实例可以耐受烯烃,烯丙基和芳环的不同取代,并可以合成三种2 H-色烯天然产物,从而证明了这种新方法。
更新日期:2017-05-19
中文翻译:

级联Claisen重排/ ö -quinone甲基化物形成/ electrocyclization方法2个ħ -chromenes
已经开发出用于合成8-取代的2 H-色烯的新方法,其特征在于新颖的级联芳族克莱森重排/邻醌甲基化物形成/6π-电环化。28个实例可以耐受烯烃,烯丙基和芳环的不同取代,并可以合成三种2 H-色烯天然产物,从而证明了这种新方法。











































 京公网安备 11010802027423号
京公网安备 11010802027423号