Abstract
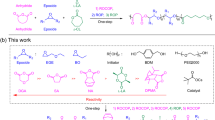
Sulfur(vi) fluoride exchange (SuFEx) is a category of click chemistry that enables covalent linking of modular units through sulfur(vi) connective hubs. The efficiency of SuFEx and the stability of the resulting bonds have led to polymer chemistry applications. Now, we report the SuFEx click chemistry synthesis of several structurally diverse SOF4-derived copolymers based on the polymerization of bis(iminosulfur oxydifluorides) and bis(aryl silyl ethers). This polymer class presents two key characteristics. First, the [–N=S(=O)F–O–] polymer backbone linkages are themselves SuFExable and undergo precise SuFEx-based post-modification with phenols or amines to yield branched functional polymers. Second, studies of individual polymer chains of several of these new materials indicate helical polymer structures. The robust nature of SuFEx click chemistry offers the potential for post-polymerization modification, enabling the synthesis of materials with control over composition and conformation.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Data supporting the findings of this study are available within the Article and its Supplementary Information. Crystallographic data for the structures reported in this Article have been deposited at the Cambridge Crystallographic Data Centre, under deposition no. CCDC 2026570 [(R)-(−)-16]. Copies of the data can be obtained free of charge via https://www.ccdc.cam.ac.uk/structures/. Source data are provided with this paper.
References
Sharpless, K. B. & Kolb, H. C. in Book of Abstracts, 217th ACS National Meeting, Anaheim, CA, March 21–25 145538 (ACS, 1999).
Kolb, H. C., Finn, M. G. & Sharpless, K. B. Click chemistry: diverse chemical function from a few good reactions. Angew. Chem. Int. Ed. 40, 2004–2021 (2001).
Tornoe, C. W., Christensen, C. & Meldal, M. Peptidotriazoles on solid phase: 1,2,3-triazoles by regiospecific copper(i)-catalyzed 1,3-dipolar cycloadditions of terminal alkynes to azides. J. Org. Chem. 67, 3057–3064 (2002).
Rostovtsev, V. V., Green, L. G., Fokin, V. V. & Sharpless, K. B. A stepwise Huisgen cycloaddition process: copper(i)-catalyzed regioselective ‘ligation’ of azides and terminal alkynes. Angew. Chem. Int. Ed. 41, 2596–2599 (2002).
Dong, J., Krasnova, L., Finn, M. G. & Sharpless, K. B. Sulfur(vi) fluoride exchange (SuFEx): another good reaction for click chemistry. Angew. Chem. Int. Ed. 53, 9430–9448 (2014).
Kolb, H. C. & Sharpless, K. B. The growing impact of click chemistry on drug discovery. Drug Discov. Today 8, 1128–1137 (2003).
Moses, J. E. & Moorhouse, A. D. The growing applications of click chemistry. Chem. Soc. Rev. 36, 1249–1262 (2007).
Moorhouse, A. D. & Moses, J. E. Click chemistry and medicinal chemistry: a case of ‘cyclo-addiction’. ChemMedChem 3, 715–723 (2008).
Thirumurugan, P., Matosiuk, D. & Jozwiak, K. Click chemistry for drug development and diverse chemical–biology applications. Chem. Rev. 113, 4905–4979 (2013).
Rouhanifard, S. H., Nordstrom, L. U., Zheng, T. & Wu, P. Chemical probing of glycans in cells and organisms. Chem. Soc. Rev. 42, 4284–4296 (2013).
McKay, C. S. & Finn, M. G. Click chemistry in complex mixtures: bioorthogonal bioconjugation. Chem. Biol. 21, 1075–1101 (2014).
Xi, W., Scott, T. F., Kloxin, C. J. & Bowman, C. N. Click chemistry in materials science. Adv. Funct. Mater. 24, 2572–2590 (2014).
Liu, Y. et al. Click chemistry in materials synthesis. III. Metal-adhesive polymers from Cu(i)-catalyzed azide–alkyne cycloaddition. J. Polym. Sci. A 45, 5182–5189 (2007).
Diaz, D. D. et al. Click chemistry in materials synthesis. 1. Adhesive polymers from copper-catalyzed azide–alkyne cycloaddition. J. Polym. Sci. A 42, 4392–4403 (2004).
Wu, P. et al. Efficiency and fidelity in a click-chemistry route to triazole dendrimers by the copper(i)-catalyzed ligation of azides and alkynes. Angew. Chem. Int. Ed. 43, 3928–3932 (2004).
van Steenis, D. J. V. C., David, O. R. P., van Strijdonck, G. P. F., van Maarseveen, J. H. & Reek, J. N. H. Click-chemistry as an efficient synthetic tool for the preparation of novel conjugated polymers. Chem. Commun. 2005, 4333–4335 (2005).
Srinivasachari, S., Liu, Y., Zhang, G., Prevette, L. & Reineke, T. M. Trehalose click polymers inhibit nanoparticle aggregation and promote pDNA delivery in serum. J. Am. Chem. Soc. 128, 8176–8184 (2006).
Gahtory, D. et al. Quantitative and orthogonal formation and reactivity of SuFEx platforms. Chem. Eur. J. 24, 10550–10556 (2018).
Randall, J. D. et al. Modification of carbon fibre surfaces by sulfur–fluoride exchange click chemistry. ChemPhysChem 19, 3176–3181 (2018).
Kassick, A. J. et al. SuFEx-based strategies for the preparation of functional particles and cation exchange resins. Chem. Commun. 55, 3891–3894 (2019).
Fan, H. et al. Sulfur(vi) fluoride exchange polymerization for large conjugate chromophores and functional main-chain polysulfates with nonvolatile memory performance. ChemPlusChem 83, 407–413 (2018).
Park, S. et al. SuFEx in metal–organic frameworks: versatile postsynthetic modification tool. ACS Appl. Mater. Interfaces 10, 33785–33789 (2018).
Li, S., Wu, P., Moses, J. E. & Sharpless, K. B. Multidimensional SuFEx click chemistry: sequential sulfur(vi) fluoride exchange connections of diverse modules launched from an SOF4 hub. Angew. Chem. Int. Ed. 56, 2903–2908 (2017).
Gao, B., Li, S., Wu, P., Moses, J. E. & Sharpless, K. B. SuFEx chemistry of thionyl tetrafluoride (SOF4) with organolithium nucleophiles: synthesis of sulfonimidoyl fluorides, sulfoximines, sulfonimidamides and sulfonimidates. Angew. Chem. Int. Ed. 57, 1939–1943 (2018).
Zheng, Q., Dong, J. & Sharpless, K. B. Ethenesulfonyl fluoride (ESF): an on-water procedure for the kilogram-scale preparation. J. Org. Chem. 81, 11360–11362 (2016).
Smedley, C. J. et al. 1-Bromoethene-1-sulfonyl fluoride (BESF) is another good connective hub for SuFEx click chemistry. Chem. Commun. 54, 6020–6023 (2018).
Leng, J. & Qin, H.-L. 1-Bromoethene-1-sulfonyl fluoride (1-Br-ESF), a new SuFEx clickable reagent, and its application for regioselective construction of 5-sulfonylfluoro isoxazoles. Chem. Commun. 54, 4477–4480 (2018).
Thomas, J. & Fokin, V. V. Regioselective synthesis of fluorosulfonyl 1,2,3-triazoles from bromovinylsulfonyl fluoride. Org. Lett. 20, 3749–3752 (2018).
Dong, J., Sharpless, K. B., Kwisnek, L., Oakdale, J. S. & Fokin, V. V. SuFEx-based synthesis of polysulfates. Angew. Chem. Int. Ed. 53, 9466–9470 (2014).
Gao, B. et al. Bifluoride-catalysed sulfur(vi) fluoride exchange reaction for the synthesis of polysulfates and polysulfonates. Nat. Chem. 9, 1083–1088 (2017).
Wang, H. et al. SuFEx-based polysulfonate formation from ethenesulfonyl fluoride-amine adducts. Angew. Chem. Int. Ed. 56, 11203–11208 (2017).
Cowie, J. M. G. & Arrighi, V. in Polymers: Chemistry and Physics of Modern Materials 3rd edn (eds Cowie, J. M. G. & Arrighi, V.) 29–56 (CRC Press, 2007).
Gauthier, M. A., Gibson, M. I. & Klok, H.-A. Synthesis of functional polymers by post-polymerization modification. Angew. Chem. Int. Ed. 48, 48–58 (2009).
Boaen, N. K. & Hillmyer, M. A. Post-polymerization functionalization of polyolefins. Chem. Soc. Rev. 34, 267–275 (2005).
Yatvin, J., Brooks, K. & Locklin, J. SuFEx on the surface: a flexible platform for postpolymerization modification of polymer brushes. Angew. Chem. Int. Ed. 54, 13370–13373 (2015).
Oakdale, J. S., Kwisnek, L. & Fokin, V. V. Selective and orthogonal post-polymerization modification using sulfur(vi) fluoride exchange (SuFEx) and copper-catalyzed azide–alkyne cycloaddition (CuAAC) reactions. Macromolecules 49, 4473–4479 (2016).
Brooks, K. et al. SuFEx postpolymerization modification kinetics and reactivity in polymer brushes. Macromolecules 51, 297–305 (2018).
Hong, Y., Lam, J. W. Y. & Tang, B. Z. Aggregation-induced emission. Chem. Soc. Rev. 40, 5361–5388 (2011).
del Amo, D. S. et al. Biocompatible copper(i) catalysts for in vivo imaging of glycans. J. Am. Chem. Soc. 132, 16893–16899 (2010).
Wang, W. et al. Sulfated ligands for the copper(i)-catalyzed azide–alkyne cycloaddition. Chem. Asian J. 6, 2796–2802 (2011).
Yashima, E., Maeda, K., Iida, H., Furusho, Y. & Nagai, K. Helical polymers: synthesis, structures and functions. Chem. Rev. 109, 6102–6211 (2009).
Nakano, T. & Okamoto, Y. Synthetic helical polymers: conformation and function. Chem. Rev. 101, 4013–4038 (2001).
BIOVIA, Materials Studio 6.0 (Dassault Systems).
Li, Y. et al. Hybrids of organic molecules and flat, oxide-free silicon: high-density monolayers, electronic properties and functionalization. Langmuir 28, 9920–9929 (2012).
Bose, K., Lech, C. J., Heddi, B. & Anh Tuan, P. High-resolution AFM structure of DNA G-wires in aqueous solution. Nat. Commun. 9, 1959 (2018).
Liang, D.-D. et al. Silicon-free SuFEx reactions of sulfonimidoyl fluorides: scope, enantioselectivity and mechanism. Angew. Chem. Int. Ed. 59, 7494–7500 (2020).
Pietschnig, R. Polymers with pendant ferrocenes. Chem. Soc. Rev. 45, 5216–5231 (2016).
Acknowledgements
We acknowledge financial support from the National Science Foundation (CHE-1610987 to K.B.S.), the NIH (R35GM139643 to P.W.), the ARC for Supporting Future Fellowship FT170100156 (J.M.), the Guangdong Natural Science Funds for Distinguished Young Scholar (2018B030306018 to S.L.), the Program for Guangdong Introducing Innovative and Entrepreneurial Teams (2017ZT07C069 to S.L.) and the Pearl River Talent Recruitment Program (2019QN01L111 to S.L.), the National Science Foundation of China (#21871208 to H.Z. and 21971260 to S.L.), King Abdulaziz University (H.D. and H.Z.), and Basic Research Project of leading technology in Jiangsu Province (BK20202012 to J.L.). Part of the work was carried out as a user project at the Molecular Foundry, supported by the Office of Science, Office of Basic Energy Sciences, of the US Department of Energy under contract no. DE-AC02-05CH11231. We thank L. Liang (Shanxi University) for assistance with X-ray single-crystal structural analyses and S. Ruggeri and B. van Lagen (Wageningen University) for detailed AFM analysis. We are grateful to J.R. Cappiello for proofreading and advice on the manuscript.
Author information
Authors and Affiliations
Contributions
K.B.S., J.M. and H.Z. supervised the work. S.L., K.B.S., P.W., J.L. and J.M. conceived and designed the syntheses of the SOF4-derived polymers. S.L., G.L., B.G., S.P.P., X.C. and H.K. performed the synthesis and characterization of the polymers. F.Z., L.M.K., Y.L. and J.L. collected and analysed the DSC and TGA data for all polymers. F.Z. collected the XRD data. S.P.P. performed molecular modelling, AFM and scanning Auger experiments. H.D. performed the SEM and TEM experiments. D.-D.L. collected the circular dichroism data. S.L., H.Z., K.B.S. and J.M. contributed to the preparation of the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
K.B.S., P.W., S.L. and B.G. are named as inventors on a patent applicant filed by The Scripps Research Institute (provisional patent application US62/427,489, filed on 29 November 2016, and international patent application no. PCT/US2017/063746).
Additional information
Peer review information Nature Chemistry thanks Jason Locklin, Jia Niu and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–17, Supplementary procedures and all characterization data, NMR spectra, HPLC chromatogram and Supplementary Table 1.
Supplementary Data
CIF file with structure factors.
Source data
Source Data Fig. 2
Transformation data of –N=SOF2 at the indicated times and calculation details.
Source Data Fig. 3
Intensity (a.u.) data (11 groups for water content vol% of 0–99%) from wavelength 370 nm to 570 nm.
Source Data Fig. 4
Figure 4c AFM profile.
Source Data Fig. 4
Figure 4f Amplitude profile.
Source Data Fig. 4
Figure 4g FFT amplitude.
Rights and permissions
About this article
Cite this article
Li, S., Li, G., Gao, B. et al. SuFExable polymers with helical structures derived from thionyl tetrafluoride. Nat. Chem. 13, 858–867 (2021). https://doi.org/10.1038/s41557-021-00726-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-021-00726-x
This article is cited by
-
Click processes orthogonal to CuAAC and SuFEx forge selectively modifiable fluorescent linkers
Nature Chemistry (2024)
-
C-SuFEx linkage of sulfonimidoyl fluorides and organotrifluoroborates
Nature Communications (2024)
-
Enantioselective sulfur(VI) fluoride exchange reaction of iminosulfur oxydifluorides
Nature Chemistry (2024)
-
Hydrogen-bonded organic framework-based bioorthogonal catalysis prevents drug metabolic inactivation
Nature Catalysis (2023)
-
A modular flow platform for sulfur(VI) fluoride exchange ligation of small molecules, peptides and proteins
Nature Synthesis (2023)