Abstract
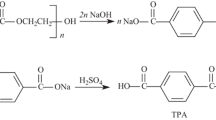
With ever increase in the consumption of bottled beverages; waste polyethylene terephthalate (wPET) bottles are rapidly emerging as a major constituent of urban waste. Amongst the various chemical techniques in vogue for recycling of wPET, glycolysis shows distinct advantage due to its ability to synthesize value added products. The current work was carried with the aim of optimizing the reaction conditions for microwave-induced catalytic glycolysis of wPET. As a first step, the effect of various cations like Zn, Ti, Fe and Co ions is reported which showed that zinc ions exhibit higher efficiency. In the second step the effect of variation in the loading of ethylene glycol (EG) glycolyzing agent showed that above a certain limit, the yield of bis-hydroxyethyl terephthalate monomer (BHET) reduced. In order to find the optimum ratio of reactants (wPET: EG) and weight of catalyst, a Taguchi design of experiments was formulated and experiments were carried. Our results show that at an optimal condition of 1:3 wPET: EG with a 1 wt% of zinc acetate gives a 65% molar yield of BHET product within 30 min.









Similar content being viewed by others
References
George N, Kurian T (2014) Recent developments in the chemical recycling of postconsumer poly(ethylene terephthalate) waste. Ind Eng Chem Res 53(37):14185–14198
Shukla SR, Harad AM, Jawale LS (2009) Chemical recycling of PET waste into hydrophobic textile dyestuffs. Polym Degrad Stab 94(4):604–609
Zahedi AR, Rafizadeh M, Taromi FA (2012) Recycling of off-grade pet via partial alcoholysis to synthesize functionalized pet oligomer nanocomposites. Polym Compos 33(10):1832–1839
Colomines G, Rivas F, Lacoste ML, Robin JJ (2005) Study of polyurethane formulations containing diols obtained via glycolysis of poly(ethylene terephthalate) (PET) by Oligoesters Diols through a reactive extrusion process. Macromol Mater Eng 290(7):710–720
Mishra S, Goje A (2003) Kinetic and thermodynamic study of methanolysis of poly(ethylene terephthalate) waste powder. Polym Int 52(3):337–342
Kurokawa H, Ohshima M, Sugiyama K, Miura H (2003) Methanolysis of polyethylene terephthalate (PET) in the presence of aluminium tiisopropoxide catalyst to form dimethyl terephthalate and ethylene glycol. Polym Degrad Stab 79(3):529–533
Davies T, Goldsmith PL, Ravens DAS, Ward IM (1962) The kinetics of the hydrolysis of polyethylene terephthalate film. J Phys Chem 66(1):175–176
Spychaj T, Fabrycy E, Spychaj S, Kacperski M (2001) Aminolysis and aminoglycolysis of waste poly(ethylene terephthalate). J Mater Cycles Waste Manag 3(1):24–31
Vaidya UR, Nadkarni VM (1989) Polyester polyols from glycolyzed PET waste: effect of glycol type on kinetics of polyesterification. J Appl Polym Sci 38(6):1179–1190
Öztürk Y, Güçlü G (2005) Unsaturated polyester resins obtained from glycolysis products of waste PET. Polym Plast Technol Eng 43(5):1539–1552
Siddiqui MN, Redhwi HH, Achilias DS (2012) Recycling of poly(ethylene terephthalate) waste through methanolic pyrolysis in a microwave reactor. J Anal Appl Pyrolysis 98:214–220
Kumagai S, Hirahashi S, Grause G, Kameda T, Toyoda H, Yoshioka T (2018) Alkaline hydrolysis of PVC-coated PET fibers for simultaneous recycling of PET and PVC. J Mater Cycles Waste Manag 20(1):439–449
Carta D, Cao G, D’Angeli C (2003) Chemical recycling of poly(ethylene terephthalate) (pet) by hydrolysis and glycolysis. Environ Sci Pollut Res 10(6):390–394
Aguado A, Martínez L, Becerra L, Arieta-araunabeña M, Arnaiz S, Asueta A et al (2014) Chemical depolymerisation of PET complex waste: hydrolysis vs. glycolysis. J Mater Cycles Waste Manag 16(2):201–210
Krzan A (1999) Poly(ethylene terephthalate) glycolysis under microwave irradiation. Polym Adv Technol 10(10):603–606
Karayannidis GP, Chatziavgoustis AP, Achilias DS (2002) Poly(ethylene terephthalate) recycling and recovery of pure terephthalic acid by alkaline hydrolysis. Adv Polym Tech 21(4):250–259
Fisher RA (1936) Design of experiments. Br Med J 1(3923):554
Liu Q, Li R, Fang T (2015) Investigating and modeling PET methanolysis under supercritical conditions by response surface methodology approach. Chem Eng J 270:535–541
Sridhar V, Shanmugharaj AM, Kim JK, Tripathy DK (2009) Optimization of carbon black and nanoclay filler loading in chlorobutyl vulcanizates using response surface methodology. Polym Compos 30(6):691–701
Li Z, Wang K, Song J, Xu Q, Kobayashi N (2014) Preparation of activated carbons from polycarbonate with chemical activation using response surface methodology. J Mater Cycles Waste Manag 16(2):359–366
Pingale ND, Shukla SR (2008) Microwave assisted ecofriendly recycling of poly (ethylene terephthalate) bottle waste. Eur Polym J 44(12):4151–4156
Chen JW, Chen LW, Cheng WH (1999) Kinetics of glycolysis of polyethylene terephthalate with zinc catalyst. Polym Int 48(9):885–888
Duque-Ingunza I, López-Fonseca R, de Rivas B, Gutiérrez-Ortiz JI (2013) Synthesis of unsaturated polyester resin from glycolysed postconsumer PET wastes. J Mater Cycles Waste Manage 15(3):256–263
Troev K, Grancharov G, Tsevi R, Gitsov I (2003) A novel catalyst for the glycolysis of poly(ethylene terephthalate). J Appl Polym Sci 90(8):148–1152
Geng Y, Dong T, Fang P, Zhou Q, Lu X, Zhang S (2015) Fast and effective glycolysis of poly(ethylene terephthalate) catalyzed by polyoxometalate. Polym Degrad Stab 117:30–36
Apicella B, Di Serio M, Fiocca L, Po R, Santacesaria E (1998) Kinetic and catalytic aspects of the formation of poly(ethylene terephthalate) (PET) investigated with model molecules. J Appl Polym Sci 69(12):2423–2433
López-Fonseca R, Duque-Ingunza I, de Rivas B, Arnaiz S, Gutiérrez-Ortiz JI (2010) Chemical recycling of post-consumer PET wastes by glycolysis in the presence of metal salts. Polym Degrad Stab 95(6):1022–1028
Pardal F, Tersac G (2006) Comparative reactivity of glycols in PET glycolysis. Polym Degrad Stab 91(11):2567–2578
Roy RK (2001) Design of experiments using the taguchi approach: 16 steps to product and process improvement. Wiley, Toronto (ISBN-10: 9780471361015)
Campanelli JR, Kamal MR, Cooper DG (1994) Kinetics of glycolysis of poly(ethylene terephthalate) melts. J Appl Polym Sci 54(11):1731–2174
Chaudhary S, Surekha P, Kumar D, Rajagopal C, Roy PK (2013) Microwave assisted glycolysis of poly(ethylene terepthalate) for preparation of polyester polyols. J Appl Polym Sci 129(5):2779–2788
Yue QF, Wang CX, Zhang LN, Ni Y, Jin YX (2011) Glycolysis of poly(ethylene terephthalate) (PET) using basic ionic liquids as catalysts. Polym Degrad Stab 96(4):399–403
Acknowledgements
This work was supported by the Industrial Strategic Technology Development Program (No. 10076439) funded by the Ministry of Trade, Industry and Energy (MOTIE, Korea) and by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. 2011-0030013, No. 2016R1A2B1013276).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Park, R., Sridhar, V. & Park, H. Taguchi method for optimization of reaction conditions in microwave glycolysis of waste PET. J Mater Cycles Waste Manag 22, 664–672 (2020). https://doi.org/10.1007/s10163-019-00958-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10163-019-00958-7