Abstract
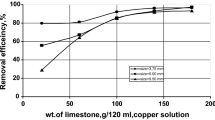
This work presents a study of sorption of heavy metals (HMs)—Cu(II), Co(II) and Cd(II)—from water media by carbonated and calcined forms of layered-double hydroxides (LDH) with various Mg(II)/Fe(III) molar ratios, which are obtained by precipitation. It is ascertained that the maximum sorption (99.9%) of the HM ions stated is observed with the use of calcined forms of LDH at \(\mathrm{pH}\,{>}\,2.8\) (pH after sorption \(\ge 8.3\)). Such an increase in the pH of the aqueous suspension causes sorption of the HM ions by the mechanism of their precipitation in the form of hydroxides or hydroxocarbonates (for Cu(II)). Sufficiently high degrees of sorption of HMs, even at low pH of the aqueous medium, are apparently caused not only by the precipitation of their hydroxide forms, but also due to the complex formation with ferrinol groups of brucite sorbent layers. An increase in the Mg/Fe ratio from 2 to 4 and a corresponding decrease in the positive charge of the layers, which determines the size of the interlayer space in the LDH, have virtually no effect on the degree of extraction of HMs. The presented results suggest that the use of the studied-LDH in practice might be promising.





Similar content being viewed by others
References
Siegel F R 2002 Environmental geochemistry of potentially toxic metals (Germany: Springer)
Nordberg G F, Fowler B A and Nordberg M 2014 Handbook on the toxicology of metals, 4th edn. (London, Waltham, San Diego: Academic Press)
Kameda T, Takeuchi H and Yoshioka T 2010 Colloids Surf. A 355 172
Liang X, Zang Y, Xu Y, Tan X, Hou W, Wang L et al 2013 Colloids Surf. A 433 122
Huang G, Wang D, Ma S, Chen J, Jiang L and Wang P 2015 J. Colloid Interface Sci. 445 294
Puzyrnaya L N, Kosorukov A A, Pshinko G N and Demchenko V Y 2014 J. Water Chem. Technol. 36 62
Milagres J L, Bellato C R, Vieira R S, Ferreira S O and Reis C 2017 J. Environ. Chem. Eng. 5 5469
Ashekuzzaman S and Jiang J 2014 Chem. Eng. J. 246 97
Zhang Y, Jing S and Liu H 2015 Chem. Eng. J. 266 21
Park M, Choi C, Seo Y J, Seo S, Choi J, Komarneni S et al 2007 Appl. Clay Sci. 37 143
Zhao D L, Sheng G D, Hu J, Chen C L and Wang X K 2011 Chem. Eng. J. 171 167
Liang X F, Hou W G and Xu J 2009 Chin. J. Chem. 27 1981
Li Y, Wang J, Li Z, Liu Q, Liu J, Liu L et al 2013 Chem. Eng. J. 218 295
Auerbach S M, Carrado K A and Dutta P K (eds) 2004 Handbook of layered materials (USA: Marcel Dekker Inc.)
Chitrakar R, Tezuka S, Hosokawa J, Makita Y, Sonoda A, Ooi K et al 2010 J. Colloid Interface Sci. 349 314
Cavani F, Trifirò F and Vaccari A 1991 Catal. Today 11 173
Ashekuzzaman S M and Jiang J-Q 2017 Process Saf. Environ. Prot. 107 454
Pshinko G N, Puzyrnaya L N, Kosorukov A A and Yatsyk B P 2017 J. Water Chem. Technol. 39 138
Vucelic M, Jones W and Moggridge G D 1997 Clays Clay Miner. 45 803
Linnik P N and Nabivanec B I 1986 Formy migracii metallov v presnyh poverhnostnyh vodah (forms of metals migration in fresh surface waters) (USSR: Gidrometeoizdat, in Russian)
Incedi J A 1979 Primeneniye kompleksov v analiticheskoj khimii (application of complexes in analytical chemistry) (USSR: Mir, in Russian)
Komarneni S, Kozai N and Roy R 1998 J. Mater. Chem. 8 1329
Pshinko G N 2013 J. Chem. 9 Article ID 347178
Lur’e J J 1971 Spravochnik po analiticheskoj khimii (handbook on analytical chemistry) (USSR: Khimija, in Russian)
Yue X, Liu W, Chen Z and Lin Z 2017 J. Environ. Sci. 53 16
Zhou H, Jiang Z and Wei S 2018 Appl. Clay Sci. 153 29
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Puzyrnaya, L.N., Pshinko, G.N., Zub, V.Y. et al. Removal of Cu(II), Co(II) and Cd(II) from water solutions by layered-double hydroxides with different [Mg(II)]/[Fe(III)] molar ratios. Bull Mater Sci 43, 3 (2020). https://doi.org/10.1007/s12034-019-1969-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12034-019-1969-z