Abstract
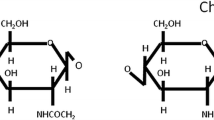
The rationale behind this study was to investigate the potential new low cost and biocompatible aqueous choline based ionic liquid for dissolution and regeneration of silk fibroin obtained from the mulberry silkworm. The silk due to its high biocompatibility and mechanical properties finds many applications in the field of biomedical science. Earlier, silk extraction methods have issues of either extraction efficiency or environmental concerns. The ionic liquid is a relatively green solvent was used to dissolve silk fibroin and optimized the process with respect to variables like temperature, time, stirring speed, type of ionic liquid and maximum dissolution ability. The dissolution process was observed through the naked eye as well as using optical microscopy. The optimized conditions at which maximum dissolution i.e. 25% was obtained, are heating the mixture at 50 °C for 2 h. Various analytical characterization such as scanning electron microscopy (SEM), Fourier transform infrared spectroscopy (FT-IR), Raman spectroscopy, X-ray diffraction (XRD), Zeta potential, nuclear magnetic resonance (1H NMR) and thermogravimetric analysis (TGA) was performed for cocoon, fiber and regenerated silk powder in order to understand the effect of ionic liquid treatment. FTIR, NMR and Raman spectra shows the characteristic peaks assigned to the silk. SEM analysis shows nanoparticles of silk fibroin powder. The crystallinity and thermal stabilities were decreased for regenerated silk as observed from XRD and TGA analysis. The nano-silk exhibited a zeta potential of − 24.6 ± 3 mV.













Similar content being viewed by others
References
Sionkowska A, Płanecka A (2013) Preparation and characterization of silk fibroin/chitosan composite sponges for tissue engineering. J Mol Liq 178:5–14
Hu X, Kaplan D, Cebe P (2006) Determining beta-sheet crystallinity in fibrous proteins by thermal analysis and infrared spectroscopy. Macromolecules 39(18):6161–6170
Shao Z, Vollrath F (2002) Materials: surprising strength of silkworm silk. Nature 418(6899):741
Zhou C-Z et al (2000) Fine organization of Bombyx mori fibroin heavy chain gene. Nucleic Acids Res 28(12):2413–2419
Yamaguchi K et al (1989) Primary structure of the silk fibroin light chain determined by cDNA sequencing and peptide analysis. J Mol Biol 210(1):127–139
Tanaka K et al (1999) Determination of the site of disulfide linkage between heavy and light chains of silk fibroin produced by Bombyx mori. Biochim Biophys Acta (BBA) 1432(1):92–103
Phillips DM et al (2004) Dissolution and regeneration of Bombyx mori silk fibroin using ionic liquids. J Am Chem Soc 126(44):14350–14351
Knowles TP et al (2007) Role of intermolecular forces in defining material properties of protein nanofibrils. Science 318(5858):1900–1903
He Y-X et al (2012) N-terminal domain of Bombyx mori fibroin mediates the assembly of silk in response to pH decrease. J Mol Biol 418(3–4):197–207
Rockwood DN et al (2011) Materials fabrication from Bombyx mori silk fibroin. Nat Protoc 6(10):1612
Passos H, Freire MG, Coutinho JA (2014) Ionic liquid solutions as extractive solvents for value-added compounds from biomass. Green Chem 16(12):4786–4815
Asim AM et al (2019) Acidic ionic liquids: promising and cost-effective solvents for processing of lignocellulosic biomass. J Mol Liq 287(1):110943
Goujon N et al (2012) Regenerated silk fibroin using protic ionic liquids solvents: towards an all-ionic-liquid process for producing silk with tunable properties. Chem Commun 48(9):1278–1280
Iqbal B et al (2017) An application of ionic liquid for preparation of homogeneous collagen and alginate hydrogels for skin dressing. J Mol Liq 243:720–725
Fox D et al (2007) The preparation and characterization of Bombyx mori silk nanocomposites using ionic liquids. ECS Trans 3(35):11–20
Xu Y et al (2005) Solubility and rheological behavior of silk fibroin (Bombyx mori) in N-methyl morpholine N-oxide. Int J Biol Macromol 35(3–4):155–161
Chen J et al (2014) What happens during natural protein fibre dissolution in ionic liquids. Materials (Basel, Switzerland) 7(9):6158–6168
Iqbal J et al (2019) COSMO-RS predictions, hydrogen bond basicity values and experimental evaluation of amino acid-based ionic liquids for lignocellulosic biomass dissolution. J Mol Liq 273:215–221
Naidu R, Zhang Y, Kim I (2017) Highly efficient green synthesis of α-hydroxyphosphonates using recyclable choline hydroxide catalyst. New J Chem 41:5373–5379
Hanley M et al (2007) Amino acid based ionic liquids: solvents for improved biopolymer dissolution. ECS Trans 3(35):41–48
Cho H-J, Um I-C (2010) The effect of dissolution condition on the yield, molecular weight, and wet-and electro-spinnability of regenerated silk fibroins prepared by LiBr aqueous solution. Int J Ind Entomol 20(2):99–105
Zhu J et al (2008) Electrospinning and rheology of regenerated Bombyx mori silk fibroin aqueous solutions: the effects of pH and concentration. Polymer 49(12):2880–2885
You R et al (2013) The degradation behavior of silk fibroin derived from different ionic liquid solvents. Nat Sci 5(06):10
Wang H-Y, Zhang Y-Q (2013) Effect of regeneration of liquid silk fibroin on its structure and characterization. Soft Matter 9(1):138–145
Whittaker JL et al (2014) Facile and rapid ruthenium mediated photo-crosslinking of Bombyx mori silk fibroin. J Mater Chem B 2(37):6259–6270
Yang R et al (2018) A novel method to prepare tussah/Bombyx mori silk fibroin-based films. RSC Adv 8(39):22069–22077
Kamalha E et al (2013) FTIR and WAXD study of regenerated silk fibroin. In: Advanced materials research. Trans Tech Publications, Pfaffikon
Frushour BG, Koenig JL (1975) Raman scattering of collagen, gelatin, and elastin. Biopolymers 14(2):379–391
Monti P et al (1998) Raman spectroscopic studies of silk fibroin from Bombyx mori. J Raman Spectrosc 29(4):297–304
Ishida M et al (1990) Solvent-induced and mechanical-treatment-induced conformational-transition of silk fibroins studied by high-resolution solid-state C-13 NMR-spectroscopy. Macromolecules 23(1):88–94
Monti P et al (2001) Raman spectroscopic characterization of Bombyx mori silk fibroin: Raman spectrum of Silk I. J Raman Spectrosc 32(2):103–107
Muhammad N et al (2017) Investigation of ionic liquids as a pretreatment solvent for extraction of collagen biopolymer from waste fish scales using COSMO-RS and experiment. J Mol Liq 232:258–264
Kim U-J et al (2005) Three-dimensional aqueous-derived biomaterial scaffolds from silk fibroin. Biomaterials 26(15):2775–2785
Ha S-W, Park YH, Hudson SM (2003) Dissolution of Bombyx mori silk fibroin in the calcium nitrate tetrahydrate–methanol system and aspects of wet spinning of fibroin solution. Biomacromolecules 4(3):488–496
Lozano-Pérez AA et al (2015) Production of silk fibroin nanoparticles using ionic liquids and high-power ultrasounds. J Appl Polym Sci 132:1–8
Murphy AR, John PS, Kaplan DL (2008) Modification of silk fibroin using diazonium coupling chemistry and the effects on hMSC proliferation and differentiation. Biomaterials 29(31):2829
Zainuddin et al (2008) The behavior of aged regenerated Bombyx mori silk fibroin solutions studied by H-1 NMR and rheology. Biomaterials 29(32):4268–4274
Kweon H, Park YH (2001) Dissolution and characterization of regenerated Antheraea pernyi silk fibroin. J Appl Polym Sci 82(3):750–758
Kweon H, Um I, Park Y (2000) Thermal behavior of regenerated Antheraea pernyi silk fibroin film treated with aqueous methanol. Polymer 41(20):7361–7367
Acknowledgements
This work has been supported by Higher Education Pakistan (NRPU No. 4099) and Interdisciplinary Research Centre in Biomedical Materials, COMSATS University Islamabad, Lahore campus, 54000, Pakistan.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Samie, M., Muhammad, N., Yameen, M.A. et al. Aqueous Solution of a Basic Ionic Liquid: A Perspective Solvent for Extraction and Regeneration of Silk Powder from Bombyx mori Silk Cocoons. J Polym Environ 28, 657–667 (2020). https://doi.org/10.1007/s10924-019-01634-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-019-01634-5