Abstract
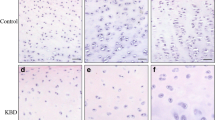
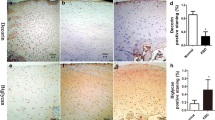
The objective of this study is to investigate the expression of enzymes involved in the sulfation of articular cartilage from proximal metacarpophalangeal (PMC) joint cartilage and distal metacarpophalangeal (DMC) joint cartilage in children with Kashin–Beck disease (KBD). The finger cartilage samples of PMC and DMC were collected from KBD and normal children aged 5–14 years old. Hematoxylin and eosin staining as well as immunohistochemical staining were used to observe the morphology and quantitate the expression of carbohydrate sulfotransferase 3 (CHST-3), carbohydrate sulfotransferase 12 (CHST-12), carbohydrate sulfotransferase 13 (CHST-13), uronyl 2-O-sulfotransferase (UST), and aggrecan. In the results, the numbers of chondrocyte decreased in all three zones of PMC and DMC in the KBD group. Less positive staining cells for CHST-3, CHST-12, CHST-13, UST, and aggrecan were observed in almost all three zones of PMC and DMC in KBD. The positive staining cell rates of CHST-12 were higher in superficial and middle zones of PMC and DMC in KBD, and a significantly higher rate of CHST-13 was observed only in superficial zone of PMC in KBD. In conclusion, the abnormal expression of chondroitin sulfate sulfotransferases in chondrocytes of KBD children may provide an explanation for the cartilage damage, and provide therapeutic targets for the treatment.







Similar content being viewed by others
References
Bayliss MT, Osborne D, Woodhouse S, Davidson C (1999) Sulfation of chondroitin sulfate in human articular cartilage. The effect of age, topographical position, and zone of cartilage on tissue composition. J Biol Chem 274:15892–15900
Cortes M, Baria AT, Schwartz NB (2009) Sulfation of chondroitin sulfate proteoglycans is necessary for proper Indian hedgehog signaling in the developing growth plate. Development 136:1697–1706
Fang H, Guo X, Farooq U, Xia C, Dong R (2012) Development and validation of a quality of life instrument for Kashin–Beck disease: an endemic osteoarthritis in China. Osteoarthr Cartil 20:630–637
Farooq U, Guo X, Chuang LH, Fang H, Zhuang GH, Xia CT (2011) Measuring health-related quality of life in Kashin–Beck disease using EQ-5D. Qual Life Res 20:425–429
Gualeni B, Facchini M, De Leonardis F, Tenni R, Cetta G, Viola M, Passi A, Superti-Furga A, Forlino A, Rossi A (2010) Defective proteoglycan sulfation of the growth plate zones causes reduced chondrocyte proliferation via an altered Indian hedgehog signalling. Matrix Biol 29:453–460. https://doi.org/10.1016/j.matbio.2010.05.001
Guo X (2011) Diagnostic, clinical and radiological characteristics of Kashin–Beck disease in Shaanxi Province, PR China. Int Orthop 25:147–150. https://doi.org/10.1007/s002640100248
Guo X, Ma W, Zhang F, Ren F, Qu C, Lammi MJ (2014) Recent advances in the research of an endemic osteochondropathy in China: Kashin–Beck disease. Osteoarthr Cartil 22:1774–1783. https://doi.org/10.1016/j.joca.2014.07.023
Guo YJ, Zhou Y, Yan SQ, Qu CJ, Wang LY, Guo X, Han J (2019) Decreased Expression of CHST-12, CHST-13, and UST in the proximal interphalangeal joint cartilage of school-age children with Kashin–Beck disease: an endemic osteoarthritis in China Caused by selenium deficiency. Biol Trace Elem Res 191:276–285
Han J, Wang W, Qu C, Liu R, Li W, Gao Z, Guo X (2015) Role of inflammation in the process of clinical Kashin-Beck disease: latest findings and interpretations. Inflamm Res 64:853–860. https://doi.org/10.1007/s00011-015-0861-6
Han J, Li D, Qu C, Wang D, Wang L, Guo X, Lammi MJ (2017) Altered expression of chondroitin sulfate structure modifying sulfotransferases in the articular cartilage from adult osteoarthritis and Kashin–Beck disease. Osteoarthr Cartil 25:1372–1375. https://doi.org/10.1016/j.joca.2017.02.803
Hiraoka N, Nakagawa H, Ong E, Akama TO, Fukuda MN, Fukuda M (2000) Molecular cloning and expression of two distinct human chondroitin 4-O-sulfotransferases that belong to the HNK-1 sulfotransferase gene family. J Biol Chem 275:20188–20196
Izumikawa T, Koike T, Kitagawa H (2012) Chondroitin 4-O-sulfotransferase-2 regulates the number of chondroitin sulfate chains initiated by chondroitin N-acetylgalactosaminyltransferase-1. Biochem J 441:697–705
Kang HG, Evers MR, Xia G, Baenziger JU, Schachner M (2002) Molecular cloning and characterization of chondroitin-4-O-sulfotransferase-3. A novel member of the HNK-1 family of sulfotransferases. J Biol Chem 277:34766
Kiani C, Chen L, Wu Y, Yee A, Yang BB (2002) Structure and function of aggrecan. Cell Res 12:19–32. https://doi.org/10.1036/sj.cr.7290106
Kobayashi M, Sugumaran G, Liu J, Shworak NW, Silbert JE, Rosenberg RD (1999) Molecular cloning and characterization of a human uronyl 2-sulfotransferase that sulfates iduronyl and glucuronyl residues in dermatan/chondroitin sulfate. J Biol Chem 274:10474–10480
Kosik-Bogacka DI, Lanocha-Arendarczyk N, Kot K, Ciosek Z, Zietek P, Karaczun M, Pilarczyk B, Tomza-Marciniak A, Podlasinska J, Kalisinska E, Pyzia J (2017) Effects of biological factors and health condition on mercury and selenium concentrations in the cartilage, meniscus and anterior cruciate ligament. J Trace Elem Med Biol 44:201–208
Kurz B, Jost B, Schünke M (2002) Dietary vitamins and selenium diminish the development of mechanically induced osteoarthritis and increase the expression of antioxidative enzymes in the knee joint of STR/1N mice. Osteoarthr Cartil 10:119–126. https://doi.org/10.1053/joca.2001.0489
Li L, Ly M, Linhardt RJ (2012a) Proteoglycan sequence. Mol BioSyst. https://doi.org/10.1039/c2mb25021g
Li S, Cao J, Caterson B, Hughes CE (2012b) Proteoglycan metabolism, cell death and Kashin–Beck disease. Glycoconj J 29:241–248. https://doi.org/10.1007/s10719-012-9421-2
Liu YQ (2018) General epidemic situation of Kashin–Beck disease and perspectives on its elimination in China. Chin J Endemiol 37:173–176 (in Chinese)
Luo M, Chen J, Li S, Sun H, Zhang Z, Fu Q, Li J, Wang J, Hughes CE, Caterson B, Cao J (2014) Changes in the metabolism of chondroitin sulfate glycosaminoglycans in articular cartilage from patients with Kashin–Beck disease. Osteoarthr Cartil 22:986–995. https://doi.org/10.1016/j.joca.2014.05.012
Ohtake S, Ito Y, Fukuta M, Habuchi O (2011) Human N-acetylgalactosamine 4-sulfate 6-O-sulfotransferase cDNA is related to human B cell recombination activating gene-associated gene. J Biol Chem 276:43894–43900
Roughley PJ, Mort JS (2014) The role of aggrecan in normal and osteoarthritic cartilage. J Exp Orthopaed 1:8. https://doi.org/10.1186/s40634-014-0008-7
Soares da Costa D, Reis RL, Pashkuleva I (2017) Sulfation of glycosaminoglycans and its implications in human health and disorders. Annu Rev Biomed Eng 19:1–26
Sobue Y, Kojima T, Kurokouchi K, Takahashi S, Yoshida H, Poole R, Ishiguro N (2017) Prediction of progression of damage to articular cartilage 2 years after anterior cruciate ligament reconstruction: use of aggrecan and type II collagen biomarkers in a retrospective observational study. Arthr Res Ther 19:265. https://doi.org/10.1186/s13075-017-1471-1
Srivastava P, Pandey H, Agarwal D, Mandal K, Phadke SR (2017) Spondyloepiphyseal dysplasia Omani type: CHST3 mutation spectrum and phenotypes in three Indian families. Am J Med Genet Part A 173:163–168
Stattin EL, Wiklund F, Lindblom K, Onnerfjord P, Jonsson BA, Tegner Y, Sasaki T, Struglics A, Lohmander S, Dahl N, Heinegård D, Aspberg A (2010) A missense mutation in the aggrecan C-type lectin domain disrupts extracellular matrix interactions and causes dominant familial osteochondritis dissecans 86:126–137
Sun M, Hussain S, Hu Y, Yan J, Min Z, Lan X, Guo Y, Zhao Y, Huang H, Feng M, Han Y, Zhang F, Zhu W, Meng L, Li D, Sun J, Lu S (2019) Maintenance of SOX9 stability and ECM homeostasis by selenium-sensitive PRMT5 in cartilage. Osteoarthr Cartil 27:932–944
Wang L, Guo X, Yi J, Qu C, Lei J, Guo Q, Han J (2018) The effects of long-term low selenium diet on the expression of CHST-3, CHST-12 and UST in knee cartilage of growing rats. J Trace Elem Med Biol 50:123–129. https://doi.org/10.1016/j.jtemb.2018.06.021
Wang L, Yin J, Yang B, Qu C, Lei J, Han J, Guo X (2019) Serious selenium deficiency in the serum of patients with Kashin–Beck disease and the effect of nano-selenium on their chondrocytes. Biol Trace Elem Res. https://doi.org/10.1007/s12011-019-01759-7
Yang Z, Xu Y, Luo HR, Ma XH, Wang Q, Wang YC, Deng W, Jiang T, Sun GQ, He TT, Hu JC, Li YR, Wang J, Li T, Hu X (2014) Whole-exome sequencing for the identification of susceptibility genes of Kashin–Beck disease. PLoS One 9:e92298
Yin PP (1979) Clinical diagnosis and differential diagnosis of Kaschin-Beck disease. Shaanxi Med J 12:40–42 (In Chinese)
Zhang F, Guo X, Zhang YP, Wen Y, Wang WZ, Wang S, Yang TL, Shen H, Chen XD, Tian Q, Tan LJ, Deng HW (2014) Genome-wide copy number variation study and gene expression analysis identify ABI3BP as a susceptibility gene for Kashin–Beck disease. Hum Genet 133:399–793
Acknowledgements
We thank Zengtie Zhang in School of Public Health, Xi’an Jiaotong University for his support and cooperation in the staining of the cartilage slices.
Funding
This study was supported by Shenzhen Science and Technology Project (JCYJ20170816100755047), the National Natural Science Foundation of China (81872567, 81402639), and China Postdoctoral Science Funding (2018M633530). The study sponsors were not involved in the study design, data collection, analysis or interpretation, or in the writing of the manuscript, neither did they affect the decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Contributions
All authors participated in drafting the article and critically modifying the important content of knowledge, with all authors endorsing the final version. Dr. Han had full access to all data in the study and takes responsibility for the integrity of the data and accuracy of the data analysis. Study conception and design: Han. Acquisition of data: Lei, Yan, Zhou. Analysis and interpretation of data: Lei, Yan, Zhou, Wang, Han, Zhang, Lammi, Qu. Drafting the article or revising it critically for important intellectual content: Lei, Yan, Lammi, Han, Qu. Final approval of the version of the article to be published: Lei, Yan, Zhou, Wang, Zhang, Guo, Lammi, Han, Qu.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest. The authors’ affiliations are shown on the cover page. The authors have sole responsibility for the writing and content of the paper.
Ethical approval
This study was approved by medical research permission from the Human Ethical Committee in Xi’an Jiaotong University. All the participants including two groups KBD (n = 5) and control (n = 5) and the legal guardians were provided with a full understanding of this study and gave written informed consent.
Antibodies used in the study
All antibodies were commercially obtained and datasheets verifying their authenticity and reactivity are available upon request, either from the authors or from the respective manufacturing/distributing companies.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lei, J., Yan, S., Zhou, Y. et al. Abnormal expression of chondroitin sulfate sulfotransferases in the articular cartilage of pediatric patients with Kashin–Beck disease. Histochem Cell Biol 153, 153–164 (2020). https://doi.org/10.1007/s00418-019-01833-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-019-01833-0