Abstract
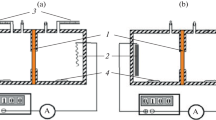
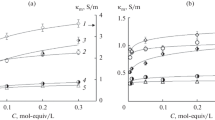
Bulk resistance and other electrochemical properties of membranes of K+-selective electrodes (ISEs) containing valinomycin are measured by means of chronopotentiometry and electrochemical impedance. It is shown that the bulk resistance of the membranes, within the Nernstian potentiometric response range, increases along decrease of KCl concentration in solution. Analogous results were reported earlier for Ca2+ and NO\(_{3}^{ - }\) ISEs. This non-constancy of the bulk resistance is in conflict with current views on the mechanism of ISEs response. Tentatively, this paradox is ascribed to heterogeneity of membranes due to water uptake from solution.







Similar content being viewed by others
REFERENCES
Morf, W.E., The Principles of Ion-Selective Electrodes and of Membrane Transport, Budapest: Akademiai Kiado, 2003.
Bakker, E., Buhlmann, P., and Pretsch, E., Chem. Rev., 1997, vol. 97, p. 3083.
Bobacka, J., Ivaska, A., and Lewenstam, A., Chem. Rev., 2008, vol. 108, p. 329.
Mikhelson, K.N., Ion-Selective Electrodes, Heidelberg, New York, Dordrecht, London: Springer, 2013.
Mikhelson, K.N. and Peshkova, M.A., Russ. Chem. Rev., 2015, vol. 84, p. 555.
Shul’ga, A.A., Ahlers, B., and Cammann, K., Electroanal. Chem. Interfacial Electrochem., 1995, vol. 395, p. 305.
Cammann, K., Ahlers, B., Henn, D., Dumschat, C., and Shul’ga, A.A., Sens. Actuat. B, 1996, vol. 35, p. 26.
Kim, Y. and Amemiya, S., Anal. Chem., 2008, vol. 80, p. 6056.
Greenawalt, P.J. and Amemiya, S., Anal. Chem., 2016, vol. 88, p. 5827.
Yuan, D., Cuartero, M., Crespo, G.A., and Bakker, E., Anal. Chem., 2017, vol. 89, p. 586.
Yuan, D., Cuartero, M., Crespo, G.A., and Bakker, E., Anal. Chem., 2017, vol. 89, p. 595.
Jarolimova, Z., Bosson, J., Labrador, G.M., Lacour, J., and Bakker, E., Electroanalysis, 2018, vol. 30, p. 650.
Vanamo, U., Hupa, E., Yrjänä, V., and Bobacka, J., Anal. Chem., 2016, vol. 88, p. 4369.
Han, T., Vanamo, U., and Bobacka, J., ChemElectroChem, 2016, vol. 3, p. 2071.
Jarolímová, Z., Han, T., Mattinen, U., Bobacka, J., and Bakker, E., Anal. Chem., 2018, vol. 90, p. 8700.
Jaworska, E., Pawlowski, P., Michalska, A., and Maksymiuk, K., Electroanalysis, 2018, vol. 30, p. 343.
Mikhelson, K.N., Electroanalysis, 2003, vol. 15, p. 1236.
Egorov, V.V., Novakovskii, A.D., and Zdrachek, E.A., Russ. J. Electrochem., 2018, vol. 54, p. 381.
Egorov, V.V., Novakovskii, A.D., and Zdrachek, E.A., Anal. Chem., 2018, vol. 90, p. 1309.
Morf, W.E., Badertscher, M., Zwickl, T., de Rooij, N.F., and Pretsch, E., J. Electroanal. Chem., 2002, vol. 526, p. 19.
Ivanova, A.D., Koltashova, E.S., Solovyeva, E.V., Peshkova, M.A., and Mikhelson, K.N., Electrochim. Acta, 2016, vol. 213, p. 439.
Sokalski, T. and Lewenstam, A., Electrochem. Commun., 2001, vol. 3, p. 107.
Kucza, W., Danielewski, M., and Lewenstam, A., Electrochem. Commun., 2006, vol. 8, p. 416.
Jasielec, J.J., Sokalski, T., Filipek, R., and Lewenstam, A., Anal. Chem., 2015, vol. 87, p. 8665.
Szyszkiewicz, K., Jasielec, J.J., Danielewski, M., and Lewenstam, A., J. Electrochem. Soc., 2017, vol. 164, p. E3559.
Shvarev, A.E., Rantsan, D.A., and Mikhelson, K.N., Sens. Actuat. B, 2001, vol. 76, p. 500.
Kondratyeva, Ye.O., Solovyeva, E.V., Khripoun, G.A., and Mikhelson, K.N., Electrochim. Acta, 2018, vol. 259, p. 458.
Ivanova, A. and Mikhelson, K., Sensors, 2018, vol. 18, p. 2062.
Mikhelson, K.N., Bobacka, J., Lewenstam, A., and Ivaska, A., Electroanalysis, 2001, vol. 13, p. 876.
Bühlmann, P., Pretsch, E., and Bakker, E., Chem. Rev., 1998, vol. 98, p. 1593.
Mikhelson, K.N., Bobacka, J., Ivaska, A., Lewenstam, A., and Bochenska, M., Anal. Chem., 2002, vol. 74, p. 518.
Mikhelson, K.N., Lutov, V.M., Sulko, K., and Stefanova, O.K., Sov. Electrochem., 1988, vol. 24, p. 1369.
Peshkova, M.A., Korobeynikov, A.I., and Mikhelson, K.N., Electrochim. Acta, 2008, vol. 53, p. 5819.
Oesch, U. and Simon, W., Helv. Chim. Acta, 1979, vol. 62, p. 754.
Bodor, S., Zook, J.M., Lindner, E., Tóth, K., and Gyurcsányi, R.E., J. Solid State Electrochem., 2009, vol. 13, p. 171.
Zook, J., Bodor, S., Lindner, E., Tóth, K., and Gyurcsányi, R.E., Electroanalysis, 2009, vol. 21, p. 1923.
Chan, A.D.C., Li, X., and Harrison, J.D., Anal. Chem., 1992, vol. 64, p. 2512.
Li, Z., Li, X., Petrovic, S., and Harrison, D.J., Anal. Chem., 1996, vol. 68, p. 1717.
Lindfors, T., Sundfors, F., Höfler, L., and Gyur-csányi, G.E., Electroanalysis, 2009, vol. 21, p. 1914.
He, N. and Lindfors, T., Anal. Chem., 2013, vol. 85, p. 1006.
ACKNOWLEDGMENTS
The authors kindly acknowledge Professor V.V. Malev for his interest in this study and stimulating discussions.
Funding
The study was performed with financial support from Russian Foundation for Basic Research, grant 19-03-00259.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare the absence of conflict of interests.
Additional information
This paper is dedicated to the 80th anniversary of Professor V.V. Malev who has made a considerable contribution into modern directions of electrochemistry.
Rights and permissions
About this article
Cite this article
Kondratyeva, Y.O., Solovyeva, E.V., Khripoun, G.A. et al. Paradox of the Variation of the Bulk Resistance of Potassium Ion-Selective Electrode Membranes within Nernstian Potentiometric Response Range. Russ J Electrochem 55, 1118–1126 (2019). https://doi.org/10.1134/S1023193519110090
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1023193519110090