Abstract
Introduction
Diabetes and cancer are among the most frequent causes of death worldwide. Recent epidemiological findings have indicated a link between diabetes and cancer in several organs, particularly the liver. A number of epidemiological studies have demonstrated that diabetes is an established independent risk factor for hepatocellular carcinoma (HCC). However, the metabolites connecting diabetes and HCC remains less well understood.
Objectives
The study aimed to identify clinical and metabolomics differences of HCC from patients with/without diabetes using comprehensive global metabolomics analysis.
Methods
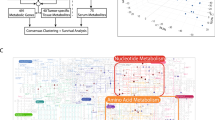
Metabolite profiling was conducted with the Metabolon platform for 120 human diabetes/non-diabetes HCC tumor/normal tissues. Standard statistical analyses were performed using the Partek Genomics Suite on log-transformed data. Principal component analysis (PCA) was conducted using all and dysregulated metabolites.
Results
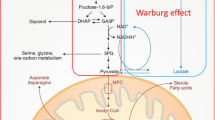
We identified a group of metabolites that are differentially expressed in the tumor tissues of diabetes HCC compared to non-diabetes HCC patients. Meanwhile, we also identified a group of metabolites that are differentially expressed in the matched normal liver tissues of diabetes HCC compared to non-diabetes HCC patients. Some metabolites are consistently dysregulated in the tumor or matched normal tissues of HCC with or without diabetes. However, some metabolites, including 2-hydroxystearate, were only overexpressed in the tumor tissues of HCC with diabetes and associated with the glucose level.
Conclusion
Metabolic profiling identifies distinct dysregulated metabolites in HCC patients with/without diabetes.





Similar content being viewed by others
Abbreviations
- HCC:
-
Hepatocellular carcinoma
- PCA:
-
Principal component analysis
- CHB:
-
Chronic hepatitis B
- HBV:
-
Hepatitis B virus
- GC–MS:
-
Gas chromatography coupled to mass spectrometry
- LTB4:
-
Leukotriene B4
- T2D:
-
Type 2 diabetes mellitus
- UPLC–MS/MS:
-
Ultra performance liquid chromatography–tandem mass spectrometer
References
Bijlsma, M. F., Sadanandam, A., Tan, P., & Vermeulen, L. (2017). Molecular subtypes in cancers of the gastrointestinal tract. Nature Reviews Gastroenterology and Hepatology, 14, 333.
Chiba, K., et al. (2017). Mutations in the promoter of the telomerase gene TERT contribute to tumorigenesis by a two-step mechanism. Science, eaao0535.
Farmer, H., et al. (2005). Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature, 434, 917.
Gan, H. K., et al. (2015). Phase I study of ABT-414 mono- or combination therapy with temozolomide (TMZ) in recurrent glioblastoma (GBM). Journal of Clinical Oncology. https://doi.org/10.1200/jco.2015.33.15_suppl.2016.
Giovannucci, E., et al. (2010). Diabetes and cancer: A consensus report. CA: A Cancer Journal for Clinicians, 60, 207–221.
Jaacks, L. M., Siegel, K. R., Gujral, U. P., & Narayan, K. V. (2016). Type 2 diabetes: A 21st century epidemic. Best Practice & Research Clinical Endocrinology & Metabolism, 30, 331–343.
Jerby-Arnon, L., et al. (2014). Predicting cancer-specific vulnerability via data-driven detection of synthetic lethality. Cell, 158, 1199–1209.
Liu, C. Y., & Feng, G.-S. (2014). NCOA5, a molecular link between type 2 diabetes and liver cancer. Hepatobiliary Surgery and Nutrition, 3, 106–108.
Lorenzo Scappaticcio, M. I. M., Bellastella, G., Giugliano, D., & Esposito, K. (2017). Insights into the relationships between diabetes, prediabetes, and cancer. Endocrine, 56(2), 231–239.
Lou, Y., Fan, F., Mu, Y., & Dong, X. (2018). The implication of diabetes metabolomics in the early diagnosis and pathogenesis of pancreatic cancer. Journal of Biological Regulators and Homeostatic Agents, 32, 75–82.
Lu, J., Xie, G., Jia, W., & Jia, W. (2013). Metabolomics in human type 2 diabetes research. Frontiers of Medicine, 7, 4–13.
Mantovani, A., & Targher, G. (2017). Type 2 diabetes mellitus and risk of hepatocellular carcinoma: Spotlight on nonalcoholic fatty liver disease. Annals of Translational Medicine. https://doi.org/10.21037/atm.2017.04.41.
Mook-Kanamori, D. O., et al. (2014). 1, 5-Anhydroglucitol in saliva is a noninvasive marker of short-term glycemic control. The Journal of Clinical Endocrinology & Metabolism, 99, E479–E483.
Osuka, S., & Van Meir, E. G. (2017). Overcoming therapeutic resistance in glioblastoma: The way forward. The Journal of Clinical Investigation, 127, 415–426.
Park, E. J., et al. (2010). Dietary and genetic obesity promote liver inflammation and tumorigenesis by enhancing IL-6 and TNF expression. Cell, 140, 197–208.
Peng, Z., Liu, C., & Wu, M. (2018). New insights into long noncoding RNAs and their roles in glioma. Molecular cancer, 17, 61.
Setiawan, V. W., et al. (2014). Diabetes and racial/ethnic differences in hepatocellular carcinoma risk: The multiethnic cohort. Journal of the National Cancer Institute. https://doi.org/10.1093/jnci/dju326.
Singh, S. K., et al. (2004). Identification of human brain tumour initiating cells. Nature, 432, 396.
Villanueva, A., & Luedde, T. (2016). The transition from inflammation to cancer in the liver. Clinical Liver Disease, 8, 89–93.
Welter, M., et al. (2018). Data for serum 1, 5 anhydroglucitol concentration in different populations. Data in Brief, 20, 753–760.
Yang, J. D., Mohamed, H. A., Cvinar, J. L., Gores, G. J., Roberts, L. R., & Kim, W. R. (2016). Diabetes mellitus heightens the risk of hepatocellular carcinoma except in patients with hepatitis C cirrhosis. The American Journal of Gastroenterology, 111, 1573–1580.
Yousri, N. A., et al. (2015). A systems view of type 2 diabetes-associated metabolic perturbations in saliva, blood and urine at different timescales of glycaemic control. Diabetologia, 58, 1855–1867.
Acknowledgements
This work was supported by grants from the National Medical Research Council of Singapore, the National Young 1000 Talents Program of China, the Jiangsu Province Education Department, the Jiangsu Province “Innovative and the Entrepreneurial Team” and “Innovative and Entrepreneurial Talent.”.
Author information
Authors and Affiliations
Contributions
HX and KMH conceived, designed and HX performed the experiments, HX, KS and JC analyzed the data and wrote the manuscript; MS provide the clinical samples and clinical data; KMH and TX sourced funding, KMH supervised the work and contributed to writing the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest concerning the work described.
Ethical approval
This study was approved by the research ethics committee of the Sun Yat-sen University Cancer Center (Guangzhou, China). All tissue samples were collected in accordance with the protocols approved by the Institutional Review Board of the Sun Yat-sen University Cancer Center, and informed consent was obtained from all patients before tissue samples were collected.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xia, H., Chen, J., Sekar, K. et al. Clinical and metabolomics analysis of hepatocellular carcinoma patients with diabetes mellitus. Metabolomics 15, 156 (2019). https://doi.org/10.1007/s11306-019-1619-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-019-1619-x