Abstract
Introduction
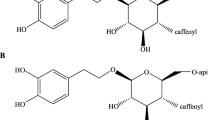
Formononetin (MBHS) and its glycosylated derivative ononin (MBHG), as the major isoflavones, have exhibited the anti-inflammatory impacts on the lipopolysaccharide (LPS)-induced inflammation. Although various researches have focused on interpreting the pharmaceutical activities of MBHG and MBHS, the molecular mechanisms in zebrafish models are still unclear.
Objective
The purpose of the present work is to investigate the molecular mechanisms of the anti-inflammatory effects of MGHG and MBHS based on lipidomics and targeted transcriptomics.
Methods
UHPLC-MS was applied for the lipid analyses and RT-PCR was adopted for the mRNA analyses, and the results of different groups were compared for exploring the significantly changed lipids and mRNAs.
Results
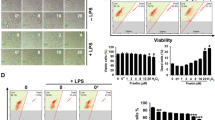
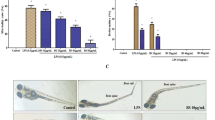
The results of lipidomics revealed that phosphatidylcholines (PCs) were drastically down-regulated in the MBHG or MBHS treated LPS-induced inflammatory zebrafish models. Besides, MBHS can also decrease the levels of triacylglycerols (TAGs). For the targeted transcriptomics analyses, 4 cytokines (TNF-α, IL-1β, IL-6 and IFN-γ) and 3 mRNA (JNK1, ERK1 and p38a) involved in the MAPK pathway were down-regulated and IL-10 was up-regulated under the treatment of MBHG or MBHS.
Conclusion
Combining the results of lipidomics and targeted transcriptomics, we indicated that MBHG and MBHS exerted potent anti-inflammatory effects on the LPS-induced zebrafish models through the MyD88 or TRIF MAPK/ERK and MAPK/JNK pathways and the glycerophospholipid, glycosylphosphatidylinositol (GPI)-anchor biosynthesis and glycerolipid metabolisms. Our results provided new insights into the anti-inflammatory mechanisms of MBHG or MBHS and supplied an effective method to interpret the pharmacological mechanisms of drugs.




Similar content being viewed by others
References
Annemarie, H. M., & Herman, P. S. (2011). Host-pathogen interactions made transparent with the zebrafish model. Current Drug Targets, 12, 1000–1017.
Bäck, M., & Hansson, G. K. (2015). Anti-inflammatory therapies for atherosclerosis. Nature Reviews Cardiology, 12, 199.
Beutler, B., & Rietschel, E. T. (2003). Innate immune sensing and its roots: the story of endotoxin. Nature Reviews Immunology, 3, 169.
Bretaud, S., Allen, C., Ingham, P. W., & Bandmann, O. (2007). p53-dependent neuronal cell death in a DJ-1-deficient zebrafish model of Parkinson’s disease. Journal of Neurochemistry, 100, 1626–1635.
Chu, A. J. (1992). Bacterial lipopolysaccharide stimulates phospholipid synthesis and phosphatidylcholine breakdown in cultured human leukemia monocytic THP-1 cells. International Journal of Biochemistry, 24, 317–323.
Clifton-Bligh, P. B., Nery, M. L., Clifton-Bligh, R. J., Visvalingam, S., Fulcher, G. R., Byth, K., et al. (2015). Red clover isoflavones enriched with formononetin lower serum LDL cholesterol-a randomized, double-blind, placebo-controlled study. European Journal of Clinical Nutrition, 69, 134–142.
Conway, E. M., Pikor, L. A., Kung, S. H., Hamilton, M. J., Lam, S., Lam, W. L., et al. (2016). Macrophages, Inflammation, and Lung Cancer. American Journal of Respiratory and Critical Care Medicine, 193, 116–130.
Dettmer, K., Aronov, P. A., & Hammock, B. D. (2007). Mass spectrometry-based metabolomics. Mass Spectrometry Reviews, 26, 51–78.
Donath, M. Y., & Shoelson, S. E. (2011). Type 2 diabetes as an inflammatory disease. Nature Reviews Immunology, 11, 98–107.
Dong, L., Yin, L., Zhang, Y., Fu, X., & Lu, J. (2017). Anti-inflammatory effects of ononin on lipopolysaccharide-stimulated RAW 264.7 cells. Molecular Immunology, 83, 46–51.
Esser, N., Legrand-Poels, S., Piette, J., Scheen, A. J., & Paquot, N. (2014). Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Research and Clinical Practice, 105, 141–150.
Grove, R. I., Allegretto, N. J., Kiener, P. A., & Warr, G. A. (1990). Lipopolysaccharide (LPS) alters phosphatidylcholine metabolism in elicited peritoneal macrophages. Journal of Leukocyte Biology, 48, 38–42.
Guan, M., Dai, D., Li, L., Wei, J., Yang, H., Li, S., et al. (2017). Comprehensive qualification and quantification of triacylglycerols with specific fatty acid chain composition in horse adipose tissue, human plasma and liver tissue. Talanta, 172, 206–214.
Hoo, R. L. C., Wong, J. Y. L., Qiao, C. F., Xu, A., Xu, H. X., & Lam, K. S. L. (2010). The effective fraction isolated from Radix Astragali alleviates glucose intolerance, insulin resistance and hypertriglyceridemia in db/db diabetic mice through its anti-inflammatory activity. Nutrition & Metabolism, 7, 67.
Lee, J. W., Mok, H. J., Lee, D. Y., Park, S. C., Kim, G. S., Lee, S. E., et al. (2017). UPLC-QqQ/MS-based lipidomics approach to characterize lipid alterations in inflammatory macrophages. Journal of Proteome Research, 16, 1460–1469.
Li, Z., Zeng, G., Zheng, X., Wang, W., Ling, Y., Tang, H., et al. (2018). Neuroprotective effect of formononetin against TBI in rats via suppressing inflammatory reaction in cortical neurons. Biomedicine & Pharmacotherapy, 106, 349–354.
Ma, Z., Ji, W., Fu, Q., & Ma, S. (2013). Formononetin inhibited the inflammation of LPS-induced acute lung injury in mice associated with induction of PPAR gamma expression. Inflammation, 36, 1560–1566.
Mu, H., Bai, Y. H., Wang, S. T., Zhu, Z. M., & Zhang, Y. W. (2009). Research on antioxidant effects and estrogenic effect of formononetin from Trifolium pratense (red clover). Phytomedicine, 16, 314–319.
Novoa, B., Bowman, T. V., Zon, L., & Figueras, A. (2009). LPS response and tolerance in the zebrafish (Danio rerio). Fish & Shellfish Immunology, 26, 326–331.
Novoa, B., & Figueras, A. (2012). Zebrafish: model for the study of inflammation and the innate immune response to infectious diseases. Advances in Experimental Medicine and Biology, 946, 253–275.
Odegaard, J. I., Ricardo-Gonzalez, R. R., Goforth, M. H., Morel, C. R., Subramanian, V., Mukundan, L., et al. (2007). Macrophage-specific PPARγ controls alternative activation and improves insulin resistance. Nature, 447, 1116.
Ouyang, W., Rutz, S., Crellin, N. K., Valdez, P. A., & Hymowitz, S. G. (2011). Regulation and functions of the IL-10 family of cytokines in inflammation and disease. Annual Review of Immunology, 29, 71–109.
Qiu, L., Ye, H., Chen, L., Hong, Y., Zhong, F., & Zhang, T. (2012). Red clover extract ameliorates dyslipidemia in streptozotocin-induced diabetic C57BL/6 mice by activating hepatic PPARalpha. Phytotherapy Research, 26, 860–864.
Rietschel, E. T., Kirikae, T., Schade, F. U., Mamat, U., Schmidt, G., Loppnow, H., et al. (1994). Bacterial endotoxin: molecular relationships of structure to activity and function. The FASEB Journal, 8, 217–225.
Takeda, K., & Akira, S. (2005). Toll-like receptors in innate immunity. International Immunology, 17, 1–14.
Xu, N., & An, J. (2017). Formononetin ameliorates mast cell-mediated allergic inflammation via inhibition of histamine release and production of pro-inflammatory cytokines. Experimental and Therapeutic Medicine, 14, 6201–6206.
Zhang, G., He, P., Tan, H., Budhu, A., Gaedcke, J., Ghadimi, B. M., et al. (2013). Integration of metabolomics and transcriptomics revealed a fatty acid network exerting growth inhibitory effects in human pancreatic cancer. Clinical Cancer Research, 19, 4983–4993.
Zhang, J., Liu, L., Wang, J., Ren, B., Zhang, L., & Li, W. (2018). Formononetin, an isoflavone from Astragalus membranaceus inhibits proliferation and metastasis of ovarian cancer cells. Journal of Ethnopharmacology, 221, 91–99.
Zhang, J.-Y., Jin, H., Wang, G.-F., Yu, P.-J., Wu, S.-Y., Zhu, Z.-G., et al. (2011). Methyl-1-hydroxy-2-naphthoate, a novel naphthol derivative, inhibits lipopolysaccharide-induced inflammatory response in macrophages via suppression of NF-κB, JNK and p38 MAPK pathways. Inflammation Research, 60, 851–859.
Acknowledgements
This work was supported by Major International S&T Cooperation Project from Ministry of Science and Technology of the People’s Republic of China (2016YFE0129000) and Fundamental Research Funds for the Central Public Welfare Research Institutes (No. ZZ10-007).
Author information
Authors and Affiliations
Contributions
LL, HZ, MF and WG designed the study. LL and JZ were responsible for doing the experiments and analyzing the data. LL wrote the article. HZ, MF and WG revised the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The experiments based on zebrafish samples were performed in accordance with the guidelines from the Association for Assessment and Accreditation of Laboratory Animal Care (AAALAC) approved by Hunter Biotechnology Co. Ltd. (Hangzhou, China) with the Approval Number as SYXK 2012-0171.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Luo, L., Zhou, J., Zhao, H. et al. The anti-inflammatory effects of formononetin and ononin on lipopolysaccharide-induced zebrafish models based on lipidomics and targeted transcriptomics. Metabolomics 15, 153 (2019). https://doi.org/10.1007/s11306-019-1614-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-019-1614-2