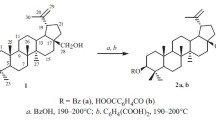
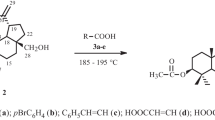
A simple and effective method for formylation of betulin using formic acid and 3a,6a-diphenylthioglycoluril is reported. The yield of betulin diformate was 87%. Allobetulin formate (95%) was prepared by reacting betulin diformate with trifluoroacetic acid. The structures of betulin diformate and allobetulin formate were confirmed by IR and NMR spectroscopy. The antioxidant activity of the synthesized compounds was studied.


Similar content being viewed by others
References
D. W. C. MacMillan, Nature (London), 455, 304 (2008).
A. G. Doyle and E. N. Jacobsen, Chem. Rev., 107, 5713 (2007).
(a) Y. Takemoto, Org. Biomol. Chem., 3, 4299 (2005); (b) P. M. Pihko, Angew. Chem., Int. Ed., 43, 2062 (2004); (c) M. S. Taylor and E. N. Jacobsen, Angew. Chem., Int. Ed., 45, 1520 (2006); (d) A. G. Doyle and E. N. Jacobsen, Chem. Rev., 107, 5713 (2007).
C. J. Broan, A. R. Butler, D. Reed, and I. H. Sadler, J. Chem. Soc., Perkin Trans. 2, 731 (1989).
L. Cao, J. Ding, G. Yin, M. Gao, M. Li, and A. Wu, Synlett, 9, 1445 (2009).
S. Khaksar, S. M. Vahdat, M. Tajbakhsh, F. Jahani, and A. Heydari, Tetrahedron Lett., 51 (49), 6388 (2010).
T. R. Shesadri and T. N. C. Vedanthan, Phytochemistry, 10, 897 (1971).
A. K. Batta and S. Rangaswami, Phytochemistry, 12, 214 (1973).
C. A. Dehelean, S. C. Pinzaru, C. I. Peev, C. Soica, and D. S. Antal, J. Optoelectron. Adv. Mater., 9, 783 (2007).
E. E. Kovac-Besovic, K. Duric, Z. Kalodera, and E. Sofic, Bosnian J. Basic Med. Sci., 9, 31 (2009).
(a) T. R. Shesadri and T. N. C. Vedanthan, Phytochemistry, 10, 897 (1971); (b) B. Green, M. D. Bentley, B. Y. Chung, N. G. Lynch, and B. L. Jensen, J. Chem. Educ., 84, 1985 (2007); (c) T.-S. Li, J.-X. Wang, and X.-J. Zheng, J. Chem. Soc., Perkin Trans. 1, 3957 (1998).
H. Schulze and K. Pieroh, “Zur Kenntnis des Betulins,” Chem. Ber., 55, 2332 (1922).
F. N. Lugemwa, F. Y. Huang, M. D. Bentley, M. J. Mendel, and A. R. A. Alford, J. Agric. Food Chem., 38, 493 (1990).
S. G. Errington, E. L. Ghisalberti, and P. R. Jefferies, Aust. J. Chem., 29, 1809 (1976).
E. Plotnikov, O. Voronova, W. Linert, D. Martemianov, E. Korotkova, E. Dorozhko, A. Astashkina, I. Martemianova, S. Ivanova, and N. Bokhan, J. App. Pharm. Sci., 6 (1), 086 (2016).
E. Plotnikov, E. Korotkova, O. Voronova, N. Sazhina, E. Petrova, A. Artamonov, L. Chernyavskaya, and E. Dorozhko, Arch. Med. Sci., 12 (5), 1071 (2016).
Acknowledgment
The work was sponsored by the Ministry of Education and Science of the Russian Federation (Grant No. 05-13108). Antioxidant activity was studied in the framework of state task “Nauka” No. 4.5752.2017 and a program for increasing the competency of Tomsk Polytechnic University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 6, November−December, 2019, pp. 941−944.
Rights and permissions
About this article
Cite this article
Arrous, S., Boudebouz, I., Parunov, I. et al. New Synthetic Method and Antioxidant Activity of Betulin Diformate and Allobetulin Formate. Chem Nat Compd 55, 1094–1097 (2019). https://doi.org/10.1007/s10600-019-02902-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-019-02902-5