Abstract
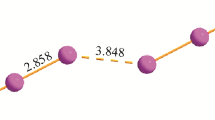
The crystal structures of tris(ethylmethylamino)cyclopropenium bromide and tris(allylmethylamino)cyclopropenium chloride are found to contain weak dimers of the cyclopropenium cation whereas the structure of the mixed salt of tris(dimethylamino)cyclopropenium chloride and dimethylammonium chloride reveals half of the cyclopropenium cations to have formed dimers and the other not to have formed dimers. These structures are compared to three related triiodide salts ([C3(NMeEt)3]I3, [C3(NEt2)3]I3 and [C3(NBu2)3]I3) as well as the previously-reported salts [C3(NEtMe)3]Cl, [C3(NH2)3]Cl, [C3(NH2)3]I and [C3(NEt2)3]I. The distances between the C3 centroids of the staggered dimers are at the short end of those normally found in π-stacked neutral arenes, let alone charged aromatic rings. Factors leading to the formation of dimers and the effect of dimer formation on the cyclopropenium structures are investigated.
Graphic Abstract
The crystal structures of triaminocyclopropenium halide salts are shown to exhibit dimer formation in some cases, but not in others, whereas corresponding triiodide salts only form cationic monomers in the solid state.



















Similar content being viewed by others
References
Hunter CA, Sanders JKM (1990) J Am Chem Soc 112:5525
Ringer AL, Sherrill CD (2009) J Am Chem Soc 131:4574
Yoshida ZI, Tawara Y (1971) J Am Chem Soc 93:2573
Komatsu K, Kitagawa T (2003) Chem Rev 103:1371
Bandar JS, Lambert TH (2013) Synthesis 45:2485
Bandar JS, Lambert TH (2013) J Am Chem Soc 135:11799
Bruns H, Patil M, Carreras J, Vázquez A, Thiel W, Goddard R, Alcarazo M (2010) Angew Chem Int Ed 49:3680
Bandar JS, Lambert TH (2012) J Am Chem Soc 134:5552
Bandar JS, Tanaset A, Lambert TH (2015) Chem Eur J 21:7365
Wilde MMD, Gravel M (2013) Angew Chem Int Ed 52:12651
Freyer JL, Brucks SD, Gobieski GS, Russell ST, Yozwiak CE, Sun M, Chen Z, Jiang Y, Bandar JS, Stockwell BR, Lambert TH, Campos LM (2016) Angew Chem Int Ed 55:12382
Kadotani S, Inagaki R, Nishihara T, Nokami T, Itoh T (2017) ACS Sustainable Chem Eng 5:8541
Mishiro K, Hu F, Paley DW, Min W, Lambert TH (2016) Eur J Org Chem 2016:1655
Curnow OJ, MacFarlane DR, Walst KJ (2011) Chem Commun 47:10248
Johnson RW (1976) Tetrahedron Lett 8:589
Jiang Y, Freyer JL, Cotanda P, Brucks SD, Killops KL, Bandar JS, Torsitano C, Balsara NP, Lambert TH, Campos LM (2015) Nat Commun 6:5950
Sevov CS, Samaroo SK, Sanford MS (2017) Adv Energy Mater 7:1602027
Weiss R, Brenner T, Hampel F, Wolski A (1995) Angew Chem Int Ed Engl 34:439
Butchard JR, Curnow OJ, Garrett DJ, Maclagan RGAR (2006) Angew Chem Int Ed 45:7550
Butchard JR, Curnow OJ, Garrett DJ, Maclagan RGAR, Libowitzky E, Piccoli PM, Schultz AJ (2012) Dalton Trans 41:11765
Weiss R, Rechinger M, Hampel F, Wolski A (1995) Angew Chem Int Ed 34:441
Weiss R, Schwab O, Hampel F (1999) Chem Eur J 5:968
Wallace AJ, Jayasinghe CD, Polson MIJ, Curnow OJ, Crittenden DL (2015) J Am Chem Soc 137:15528
Janiak C (2000) J Chem Soc Dalton Trans 21:3885
Martinez CR, Iverson BL (2012) Chem Sci 3:2191
Amabilino DB, Stoddart JF (1995) Chem Rev 95:2725
Wheeler SE, Houk KN (2008) J Am Chem Soc 130:10854
Tobey SW, West R (1966) J Am Chem Soc 88:2478
Lucier JJ, Harris AD, Korosec PS (1964) Org Synth 44:72
Wawzonek S, McKillip W, Peterson C (1964) Org Synth 44:75
Leffler MT (1938) Org Synth 18:5
Walst KJ, Yunis R, Bayley PM, MacFarlane DR, Ward CJ, Wang R, Curnow OJ (2015) RSC Adv 5:39565
Dolomanov OV, Bourhis LJ, Gildea RJ, Howard JAK, Puschmann H (2009) J Appl Cryst 42:339
Sheldrick GM (2008) Acta Cryst A64:112
Vu PD, Boydston AJ, Bielawski CW (2007) Green Chem 9:1158
Butchard JR, Curnow OJ, Pipal RJ, Robinson WT, Shang R (2008) J Phys Org Chem 21:127
Clark GR, Surman PWJ, Taylor MJ (1995) J Chem Soc Faraday Trans 91:1523
Acknowledgements
Dr. Deborah Crittenden, University of Canterbury, is thanked for useful discussions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Curnow, O.J., Jayasinghe, C.D., Polson, M.I.J. et al. Triaminocyclopropenium Halide and Triiodide Salts: The Formation of Cyclopropenium Dimers. J Chem Crystallogr 50, 357–372 (2020). https://doi.org/10.1007/s10870-019-00809-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-019-00809-1