Abstract
Background and aims
Plant litter decomposition is a crucial pathway of the global carbon (C) cycle, and controls the net primary productivity in terrestrial ecosystems. Our understanding of litter decomposition is primarily based on decay patterns observed for leaf litter tissues. The distal shoot systems (leaves and the first few orders of twigs) vary vastly in function and tissue chemistry and both decomposition patterns and controlling factors of these multi-type litter complexes are poorly understood.
Methods
We followed decomposition of leaf litter and the first three orders of twigs across 10 temperate tree species over 2 years in Northeast China.
Results
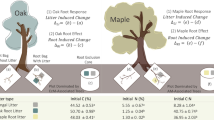
Eight species showed slower decomposition rates in twigs than leaf litter, and almost all species showed that decomposition rates decrease from first- to third-order twigs. The decomposition of twigs and leaf litter in 10 tree species were not coordinated. Most importantly, in contrast to leaf litter, the variation in twigs decomposition cannot be predicted by the commonly used parameters like N contents, but is predicted by initial concentrations of Mg and cellulose plus lignin/P ratio in twig tissues.
Conclusions
We conclude that there were significant differences in decomposition rates among the tree distal shoot systems. Leaf litter decomposition does not mirror twigs decomposition, and variation in their decomposition rates was driven by two different subset of litter traits, at least in the ten species we studied. Additionally, the heterogeneity within the tree distal shoot systems could lead to differences in their contribution to soil in term of carbon dynamics.




Similar content being viewed by others
Data availability
The datasets generated during and nalysed during the current study are available from the corresponding author on reasonable request.
References
Aerts R (1997) Climate, leaf litter chemistry and leaf litter decomposition in terrestrial ecosystems: a triangular relationship. Oikos 79:439–449. https://doi.org/10.2307/3546886
Berg B, McClaugherty C (2008) Plant litter–decomposition, humus formation, carbon sequestration (2nd edition). Springer-Verlag Berlin, Heidelberg
Berg B, McClaugherty C (2020) Climate gradients. Substrate quality versus climate and their interactions. Plant Litter: decomposition, Humus formation, Carbon Sequestration. Springer International Publishing, Cham, pp 165–187
Berg B, Wessen B, Ekbohm G (1982) Nitrogen level and lignin decomposition in scots pine needle litter. Oikos 38:291–296
Bradford MA, Berg B, Maynard DS, Wieder WR, Wood SA, Cornwell W (2016) Understanding the dominant controls on litter decomposition. J Ecol 104:229–238. https://doi.org/10.1111/1365-2745.12507
Bradford MA, Veen GFC, Bonis A, Bradford EM, Classen AT, Cornelissen JHC, Crowther TW, De Long JR, Freschet GT, Kardol P, Manrubia-Freixa M, Maynard DS, Newman GS, Logtestijn RSP, Viketoft M, Wardle DA, Wieder WR, Wood SA, van der Putten WH (2017) A test of the hierarchical model of litter decomposition. Nat Ecol Evol 1:1836–1845. https://doi.org/10.1038/s41559-017-0367-4
Brovkin V, van Bodegom PM, Kleinen T, Wirth C, Cornwell WK, Cornelissen JHC, Kattge J (2012) Plant-driven variation in decomposition rates improves projections of global litter stock distribution. Biogeosciences 9:565–576. https://doi.org/10.5194/bg-9-565-2012
Buck-Sorlin GH, Bell AD (1998) A quantification of shoot shedding in pedunculate oak (Quercus robur L). Bot J Linn Soc 127:371–391. https://doi.org/10.1006/bojl.1997.0166
Cornwell WK, Cornelissen JH, Amatangelo K, Dorrepaal E, Eviner VT, Godoy O, Hobbie SE, Hoorens B, Kurokawa H, Perez-Harguindeguy N, Quested HM, Santiago LS, Wardle DA, Wright IJ, Aerts R, Allison SD, van Bodegom P, Brovkin V, Chatain A, Callaghan TV, Diaz S, Garnier E, Gurvich DE, Kazakou E, Klein JA, Read J, Reich PB, Soudzilovskaia NA, Vaieretti MV, Westoby M (2008) Plant species traits are the predominant control on litter decomposition rates within biomes worldwide. Ecol Lett 11:1065–1071. https://doi.org/10.1111/j.1461-0248.2008.01219.x
Cornwell WK, Cornelissen JHC, Allison SD, Bauhus J, Eggleton P, Preston CM, Scarff F, Weedon JT, Wirth C, Zanne AE (2009) Plant traits and wood fates across the globe: rotted, burned, or consumed? Global Change Biol 15:2431–2449. https://doi.org/10.1111/j.1365-2486.2009.01916.x
Eldhuset TD, Kjonaas OJ, Lange H (2017) Decomposition rates and nutrient dynamics of Picea abies needles, twigs and fine roots after stem-only harvesting in eastern and western Norway. Plant Soil 418:357–375. https://doi.org/10.1007/s11104-017-3302-1
Fang M, Liang M, Liu X, Li W, Huang E, Yu S (2020) Abundance of saprotrophic fungi determines decomposition rates of leaf litter from arbuscular mycorrhizal and ectomycorrhizal trees in a subtropical forest. Soil Biol Biochem 149:107966. https://doi.org/10.1016/j.soilbio.2020.107966
Freschet GT, Cornwell WK, Wardle DA, Elumeeva TG, Liu W, Jackson BG, Onipchenko VG, Soudzilovskaia NA, Tao J, Cornelissen JHC, Austin A (2013) Linking litter decomposition of above- and below-ground organs to plant-soil feedbacks worldwide. J Ecol 101:943–952. https://doi.org/10.1111/1365-2745.12092
Guo C, Tuo B, Ci H, Sai BL, Zhang Y, Yan ER, Cornelissen JHC (2022) How detritivores, plant traits and time modulate coupling of leaf versus woody litter decomposition rates across species. J Ecol 111:227–239. https://doi.org/10.1111/1365-2745.14028
Hall SJ, Huang W, Timokhin VI, Hammel KE (2020) Lignin lags, leads, or limits the decomposition of litter and soil organic carbon. Ecology 101:e03113. https://doi.org/10.1002/ecy.3113
Jackson BG, Peltzer DA, Wardle DA (2013) Are functional traits and litter decomposability coordinated across leaves, twigs and wood? A test using temperate rainforest tree species. Oikos 122:1131–1142. https://doi.org/10.1111/j.1600-0706.2012.00056.x
Joly F-X, Milcu A, Scherer-Lorenzen M, Jean L-K, Bussotti F, Dawud SM, Mueller S, Pollastrini M, Raulund-Rasmussen K, Vesterdal L, Haettenschwiler S (2017) Tree species diversity affects decomposition through modified micro-environmental conditions across European forests. New Phytol 214:1281–1293. https://doi.org/10.1111/nph.14452
Keiblinger KM, Schneider T, Roschitzki B, Schmid E, Eberl L, Haemmerle I, Leitner S, Richter A, Wanek W, Riedel K, Zechmeister-Boltenstern S (2012) Effects of stoichiometry and temperature perturbations on beech leaf litter decomposition, enzyme activities and protein expression. Biogeosciences 9:4537–4551. https://doi.org/10.5194/bg-9-4537-2012
Kuzyakov Y (2010) Priming effects: interactions between living and dead organic matter. Soil Biol Biochem 42:1363–1371. https://doi.org/10.1016/j.soilbio.2010.04.003
Leitner S, Wanek W, Wild B, Haemmerle I, Kohl L, Keiblinger KM, Zechmeister-Boltenstern S, Richter A (2012) Influence of litter chemistry and stoichiometry on glucan depolymerization during decomposition of beech (Fagus sylvatica L.) litter. Soil Biol Biochem 50:174–187. https://doi.org/10.1016/j.soilbio.2012.03.012
Li M-H, Xiao W-F, Wang S-G, Cheng G-W, Cherubini P, Cai X-H, Liu X-L, Wang X-D, Zhu W-Z (2008) Mobile carbohydrates in Himalayan treeline trees I. evidence for carbon gain limitation but not for growth limitation. Tree Physiol 28:1287–1296. https://doi.org/10.1093/treephys/28.8.1287
Li A, Guo D, Wang Z, Liu H (2010) Nitrogen and phosphorus allocation in leaves, twigs, and fine roots across 49 temperate, subtropical and tropical tree species: a hierarchical pattern. Funct Ecol 24:224–232. https://doi.org/10.1111/j.1365-2435.2009.01603.x
Makkonen M, Berg MP, Handa IT, Hattenschwiler S, van Ruijven J, van Bodegom PM, Aerts R (2012) Highly consistent effects of plant litter identity and functional traits on decomposition across a latitudinal gradient. Ecol Lett 15:1033–1041. https://doi.org/10.1111/j.1461-0248.2012.01826.x
Melillo JM, Aber JD, Muratore JF (1982) Nitrogen and lignin control of hardwood leaf litter decomposition dynamics. Ecology 63:621–626. https://doi.org/10.2307/1936780
Olson JS (1963) Energy storage and the balance of producers and decomposers in ecological systems. Ecology 44:322–331. https://doi.org/10.2307/1932179
Pang Y, Tian J, Lv X, Wang R, Wang D, Zhang F (2022) Contrasting dynamics and factor controls in leaf compared with different-diameter fine root litter decomposition in secondary forests in the Qinling Mountains after 5 years of whole-tree harvesting. Sci Total Environ 838:156194. https://doi.org/10.1016/j.scitotenv.2022.156194
Pietsch KA, Ogle K, Cornelissen JHC, Cornwell WK, Bönisch G, Craine JM, Jackson BG, Kattge J, Peltzer DA, Penuelas J, Reich PB, Wardle DA, Weedon JT, Wright IJ, Zanne AE, Wirth C (2014) Global relationship of wood and leaf litter decomposability: the role of functional traits within and across plant organs. Global Ecol Biogeogr 23:1046–1057. https://doi.org/10.1111/geb.12172
Pregitzer KS, DeForest JL, Burton AJ, Allen MF, Ruess RW, Hendrick RL (2002) Fine root architecture of nine north American trees. Ecol Monogr 72:293–309. https://doi.org/10.1890/0012-9615(2002)072[0293:Fraonn]2.0.Co;2
Prescott CE (2010) Litter decomposition: what controls it and how can we alter it to sequester more carbon in forest soils? Biogeochemistry 101:133–149. https://doi.org/10.1007/s10533-010-9439-0
Rahman MM, Tsukamoto J, Rahman MM, Yoneyama A, Mostafa KM (2013) Lignin and its effects on litter decomposition in forest ecosystems. Chem Ecol 29:540–553. https://doi.org/10.1080/02757540.2013.790380
Schleuss PM, Widdig M, Biederman LA, Borer ET, Crawley MJ, Kirkman KP, Seabloom EW, Wragg PD, Spohn M (2021) Microbial substrate stoichiometry governs nutrient effects on nitrogen cycling in grassland soils. Soil Biol Biochem 155:108168. https://doi.org/10.1016/j.soilbio.2021.108168
Seyfried GS, Dalling JW, Yang WH (2021) Mycorrhizal type effects on leaf litter decomposition depend on litter quality and environmental context. Biogeochemistry 155:21–38. https://doi.org/10.1007/s10533-021-00810-x
Sun T, Dong L, Wang Z, Lü X, Mao Z (2016) Effects of long-term nitrogen deposition on fine root decomposition and its extracellular enzyme activities in temperate forests. Soil Biol Biochem 93:50–59. https://doi.org/10.1016/j.soilbio.2015.10.023
Sun T, Hobbie SE, Berg B, Zhang H, Wang Q, Wang Z, Hättenschwiler S (2018) Contrasting dynamics and trait controls in first-order root compared with leaf litter decomposition. Proc Natl Acad Sci U S A 115:10392–10397. https://doi.org/10.1073/pnas.1716595115
Swift MJ, Heal OW, Anderson JMJSE (1979) Decomposition in terrestrial ecosystems. University of California Press, Berkeley
Talbot JM, Treseder KK (2012) Interactions among lignin, cellulose, and nitrogen drive litter chemistry-decay relationships. Ecology 93:345–354. https://doi.org/10.1890/11-0843.1
van Soest PJ, Wine RH (1967) Use of detergents in analysis of fibrous feeds. IV. Determination of plant cell-wall constituents. J Assoc Off Anal Chem 50:50–55
Wang CG, Zheng XB, Wang AZ, Dai GH, Zhu BK, Zhao YM, Dong SJ, Zu WZ, Wang W, Zheng YG, Li JG, Li MH (2021) Temperature and precipitation diversely control seasonal and annual dynamics of litterfall in a temperate mixed mature forest, revealed by long-term data analysis. J Geophys Res -Biogeosci 126:e2020JG006204. https://doi.org/10.1029/2020JG006204
Weedon JT, Cornwell WK, Cornelissen JH, Zanne AE, Wirth C, Coomes DA (2009) Global meta-analysis of wood decomposition rates: a role for trait variation among tree species? Ecol Lett 12:45–56. https://doi.org/10.1111/j.1461-0248.2008.01259.x
Xiong Y, Fan P, Fu S, Zeng H, Guo D (2012) Slow decomposition and limited nitrogen release by lower order roots in eight Chinese temperate and subtropical trees. Plant Soil 363:19–31. https://doi.org/10.1007/s11104-012-1290-8
Yang K, Zhu J, Zhang W, Zhang Q, Lu D, Zhang Y, Zheng X, Xu S, Wang GG (2022) Litter decomposition and nutrient release from monospecific and mixed litters: comparisons of litter quality, fauna and decomposition site effects. J Ecol 110:1673–1686. https://doi.org/10.1111/1365-2745.13902
Zanne AE, Oberle B, Dunham KM, Milo AM, Walton ML, Young DF (2015) A deteriorating state of affairs: how endogenous and exogenous factors determine plant decay rates. J Ecol 103:1421–1431. https://doi.org/10.1111/1365-2745.12474
Zhou S, Butenschoen O, Barantal S, Handa IT, Makkonen M, Vos V, Aerts R, Berg MP, McKie B, Van Ruijven J, Hattenschwiler S, Scheu S (2020) Decomposition of leaf litter mixtures across biomes: the role of litter identity, diversity and soil fauna. J Ecol 108:2283–2297. https://doi.org/10.1111/1365-2745.13452
Zuo J, Hefting MM, Berg MP, van Logtestijn RSP, van Hal J, Goudzwaard L, Liu J-C, Sass-Klaassen U, Sterck FJ, Poorter L, Cornelissen JHC (2018) Is there a tree economics spectrum of decomposability? Soil Biol Biochem 119:135–142. https://doi.org/10.1016/j.soilbio.2018.01.019
Acknowledgements
We thank Hongguang Zhang, Yu Zhu, Rong Zhao and Fuqiang Long for assistance in the field and laboratory. Thanks to Siyuan Wang for constructive comments on earlier drafts of the manuscript.
Funding
The funding for this research was supported by the National Key R&D Program of China (2020YFA0608100 and 2022YFD2201300), National Natural Science Foundation of China (32022054 and 32192432), International Partnership Program of Chinese Academy of Sciences (151221KYSB20210005), Key Research Program of Frontier Sciences of Chinese Academy of Sciences (ZDBS-LY-DQC019), and the Strategic Priority Research Program of Chinese Academy of Sciences (Grant No. XDA28120100).
Author information
Authors and Affiliations
Contributions
Weiping Gu performed the data analysis and wrote the manuscript; Björn Berg performed specifically critical review, commentary and revision; Lili Dong performed the design of methodology; Fengjian Yang performed the oversight and leadership responsibility for the research activity planning and execution; Tao Sun Provided the financial support for the project leading to this publication.
Corresponding authors
Ethics declarations
Competing interest
The authors declare that they have no competing interest.
Additional information
Responsible Editor: Timothy J. Fahey.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 74.4 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gu, W., Berg, B., Dong, L. et al. Patterns and controlling factors of decomposition in distal shoot systems by branch order across 10 temperate tree species. Plant Soil (2024). https://doi.org/10.1007/s11104-024-06590-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11104-024-06590-1