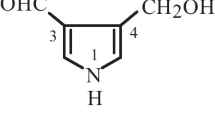
Further study on a 95% EtOH extract of the red mold rice fermented with the mutant of the fungus Monascus sp. led to the isolation of one new natural pyranoid derivative, namely monascuslactone (1), together with three known compounds. The structure of the new compound was determined as (4S*,6S*)-4-hydroxy-4,6-dimethyltetrahydro-2H-pyran-2-one. The known compounds were identified as monascin (2), ankaflavin (3), and methyl N-methyl anthranylate (4). Their structures were elucidated by 1D and 2D NMR spectroscopy together with HR-ESI-MS analysis, and comparison of the spectroscopic data with those reported for structurally related compounds.

Similar content being viewed by others
References
Z. Huang, Y. Xu, L. Li, and Y. Li, J. Agric. Food Chem., 56, 112 (2008).
J. Ma, Y. Li, Q. Ye, J. Li, Y. Hua, D. Ju, D. Zhang, R. Cooper, and M. Chang, J. Agric. Food Chem., 48, 5220 (2000).
D. Wild, G. Toth, and H. U. Humpf, J. Agric. Food Chem., 50, 3999 (2002).
P. J. Blanc, M. O. Loret, and G. Goma, Biotechnol. Lett., 17, 291 (1995).
T. Akihisa, S. Mafune, M. Ukiya, Y. Kimura, K. Yasukawa, T. Suzuki, H. Tokuda, N. Tanabe, and T. Fukuoka, J. Nat. Prod., 67, 479 (2004).
T. Akihisa, H. Tokuda, K. Yasukawa, M. Ukiya, A. Kiyota, N. Sakamoto, T. Suzuki, N. Tanabe, and H. Nishino, J. Agric. Food Chem., 53, 562 (2005).
S. Jongrungruangchok, P. Kittakoop, B. Yongsmith, R. Bavovada, S. Tanasupawat, N. Lartpornmatulee, and Y. Thebtaranonth, Phytochemistry, 65, 2569 (2004).
P. Juzlova, L. Martinkova, and V. Kren, J. Industr. Microbiol., 16, 163 (1996).
P. Juzlova, T. Rezanka, L. Martinkova, and V. Kren, Phytochemistry, 43, 151 (1996).
H. Nozaki, S. Date, H. Kondo, H. Kiyohara, D. Takaoka, T. Tada, and M. Nakayama, Agric. Biolog. Chem., 55, 899 (1991).
K. Sato, Y. Goda, S. S. Sakamoto, H. Shibata, T. Maitani, and T. Yamada, Chem. Pharm. Bull., 45, 227 (1997).
D. Wild, G. Toth, and H. U. Humpf, J. Agric. Food Chem., 51, 5493 (2003).
M. J. Cheng, M. D. Wu, I. S. Chen, and G. F. Yuan, Chem. Pharm. Bull., 56, 394 (2008).
M. Karina, Szymulanska-Ramamurthy, M. Che, and C.-T. Zhao, Acta Pharm. Sin. B, 7, 485 (2017).
C. Moulineau and J. E. Dubois, Bull. Soc. Chim. Fr., 107, 4363 (1970).
L. L. Li, J. P. Chen, and L. Y. Kong, Chin. J. Nat. Med., 4, 32 (2006).
H. Mayer and H. A. Ruttimann, Helv. Chim. Acta, 63, 1451 (1980).
Acknowledgment
This work was kindly supported by grants from the Ministry of Science and Technology, R.O.C. (MOST-108-2320-B-080-002-, MOST-109- 2622-E-080-001-, MOST-110-2320-B-080-001-, and NSTC-111-2320-B-030-010-MY3). The authors thank Senior Technician Mrs. Chyi Jia Wang of the Center for Resources, Research, and Development (CRRD) of Kaohsiung Medical University for measuring the 2D NMR data.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Khimiya Prirodnykh Soedinenii, No. 1, January–February, 2024, pp. 27–29.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, MD., Chen, MH., Chen, JJ. et al. Constituents Isolated from the Fungus Monascus sp.. Chem Nat Compd 60, 30–33 (2024). https://doi.org/10.1007/s10600-024-04245-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-024-04245-2