Abstract
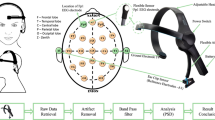
Microstate sequences summarize the changing voltage patterns measured by electroencephalography, using a clustering approach to reduce the high dimensionality of the underlying data. A common approach is to restrict the pattern matching step to local maxima of the global field power (GFP) and to interpolate the microstate fit in between. In this study, we investigate how the anesthetic propofol affects microstate sequence periodicity and predictability, and how these metrics are changed by interpolation. We performed two frequency analyses on microstate sequences, one based on time-lagged mutual information, the other based on Fourier transform methodology, and quantified the effects of interpolation. Resting-state microstate sequences had a 20 Hz frequency peak related to dominant 10 Hz (alpha) rhythms, and the Fourier approach demonstrated that all five microstate classes followed this frequency. The 20 Hz periodicity was reversibly attenuated under moderate propofol sedation, as shown by mutual information and Fourier analysis. Characteristic microstate frequencies could only be observed in non-interpolated microstate sequences and were masked by smoothing effects of interpolation. Information-theoretic analysis revealed faster microstate dynamics and larger entropy rates under propofol, whereas Shannon entropy did not change significantly. In moderate sedation, active information storage decreased for non-interpolated sequences. Signatures of non-equilibrium dynamics were observed in non-interpolated sequences, but no changes were observed between sedation levels. All changes occurred while subjects were able to perform an auditory perception task. In summary, we show that low dose propofol reversibly increases the randomness of microstate sequences and attenuates microstate oscillations without correlation to cognitive task performance. Microstate dynamics between GFP peaks reflect physiological processes that are not accessible in interpolated sequences.







Similar content being viewed by others
References
Akeju O, Pavone KJ, Westover MB, Vazquez R, Prerau MJ, Harrell PG, Hartnack KE, Rhee J, Sampson AL, Habeeb K, Gao L, Pierce ET, Walsh JL, Brown EN, Purdon PL (2014) A comparison of Propofol- and dexmedetomidine-induced Electroencephalogram Dynamics Using Spectral and coherence analysis. Anesthesiology 121(5):978–989. https://doi.org/10.1097/ALN.0000000000000419
Artoni F, Maillard J, Britz J, Seeber M, Lysakowski C, Bréchet L, Tramèr MR, Michel CM (2022) EEG microstate dynamics indicate a U-shaped path to propofol-induced loss of consciousness. NeuroImage. https://doi.org/10.1016/j.neuroimage.2022.119156
Bréchet L, Michel CM (2022) EEG microstates in altered states of consciousness. Front Psychol 13:1549. https://doi.org/10.3389/FPSYG.2022.856697/BIBTEX
Brodbeck V, Kuhn A, von Wegner F, Morzelewski A, Tagliazucchi E, Borisov S, Michel CM, Laufs H (2012) EEG microstates of wakefulness and NREM sleep. NeuroImage 62(3):2129–2139. https://doi.org/10.1016/j.neuroimage.2012.05.060
Chennu S, O’Connor S, Adapa R, Menon DK, Bekinschtein TA (2016) Brain connectivity dissociates responsiveness from drug exposure during Propofol-Induced transitions of consciousness. PLoS Comput Biol 12(1):1–17. https://doi.org/10.1371/journal.pcbi.1004669
Ching S, Brown EN (2014) Modeling the dynamical effects of anesthesia on brain circuits. Curr Opin Neurobiol 25:116–122. https://doi.org/10.1016/j.conb.2013.12.011
Custo A, Van De Ville D, Wells WM, Tomescu MI, Brunet D, Michel CM (2017) Electroencephalographic resting-state networks: source localization of Microstates. Brain Connect 7(10):671–682. https://doi.org/10.1089/brain.2016.0476
de la Fuente LA, Zamberlan F, Bocaccio H, Kringelbach M, Deco G, Perl YS, Pallavicini C, Tagliazucchi E (2023) Temporal irreversibility of neural dynamics as a signature of consciousness. Cereb Cortex 33(5):1856–1865. https://doi.org/10.1093/cercor/bhac177
Deco G, Jirsa VK, McIntosh AR (2011) Emerging concepts for the dynamical organization of resting-state activity in the brain. Nat Reviews Neurosci 12(1):43–56. https://doi.org/10.1038/nrn2961
Hao Z, Zhai X, Cheng D, Pan Y, Dou W (2022) EEG microstate-specific functional connectivity and stroke-related alterations in brain dynamics. Front NeuroSci. https://doi.org/10.3389/fnins.2022.848737
Iber C, Ancoli-Israel S, Chesson A, Quan SF (2007) The AASM manual for the scoring of sleep and associated events: rules, terminology and technical specifications, 1st edn. American Academy of Sleep Medicine, Darien
Jia W, von Wegner F, Zhao M, Zeng Y (2021) Network oscillations imply the highest cognitive workload and lowest cognitive control during idea generation in open-ended creation tasks. Sci Rep. https://doi.org/10.1038/s41598-021-03577-1
Kales A, Rechtschaffen A (1968). A manual of standardized terminology, techniques and scoring system for sleep stages of human subjects, vol 204. Government Printing Office, Washington DC
Kallionpää RE, Valli K, Scheinin A, Långsjö J, Maksimow A, Vahlberg T, Revonsuo A, Scheinin H, Mashour GA, Li D (2020) Alpha band frontal connectivity is a state-specific electroencephalographic correlate of unresponsiveness during exposure to dexmedetomidine and propofol. Br J Anaesth 125(4):518–528. https://doi.org/10.1016/j.bja.2020.05.068
Khanna A, Pascual-Leone A, Michel CM, Farzan F (2015) Microstates in resting-state EEG: current status and future directions. Neurosci Biobehavioral Reviews 49:105–113. https://doi.org/10.1016/j.neubiorev.2014.12.010
Kikuchi M, Koenig T, Wada Y, Higashima M, Koshino Y, Strik W, Dierks T (2007) Native EEG and treatment effects in neuroleptic-naïve Schizophrenic patients: time and frequency domain approaches. Schizophr Res 97(1–3):163–172. https://doi.org/10.1016/j.schres.2007.07.012
Kim H, Moon JY, Mashour GA, Lee UC (2018) Mechanisms of hysteresis in human brain networks during transitions of consciousness and unconsciousness: Theoretical principles and empirical evidence. PLoS Comput Biol. https://doi.org/10.1371/JOURNAL.PCBI.1006424
Koenig T, Valdés-Sosa PA (2018) Results obtained by combining different estimators of EEG connectivity become uninterpretable if the underlying models are incompatible. Brain connectivity 8(2):57–59. https://doi.org/10.1089/BRAIN.2017.0555
Koenig T, Lehmann D, Merlo MCG, Kochi K, Hell D, Koukkou M (1999) A deviant EEG brain microstate in acute, neuroleptic-naive schizophrenics at rest. Eur Arch Psychiatry Clin NeuroSci 249(4):205–211. https://doi.org/10.1007/s004060050088
Koenig T, Prichep L, Lehmann D, Sosa PV, Braeker E, Kleinlogel H, Isenhart R, John ER (2002) Millisecond by Millisecond, Year by Year: normative EEG microstates and Developmental stages. NeuroImage 16:41–48. https://doi.org/10.1006/nimg.2002.1070
Krylova M, Alizadeh S, Izyurov I, Teckentrup V, Chang C, van der Meer J, Erb M, Kroemer N, Koenig T, Walter M, Jamalabadi H (2021) Evidence for modulation of EEG microstate sequence by vigilance level. NeuroImage 224:117393. https://doi.org/10.1016/J.NEUROIMAGE.2020.117393
Kuhn A, Brodbeck V, Tagliazucchi E, Morzelewski A, von Wegner F, Laufs H (2015) Narcoleptic patients show fragmented EEG-Microstructure during early NREM sleep. Brain Topogr 28(4):619–635. https://doi.org/10.1007/s10548-014-0387-1
Lapointe AP, Li D, Hudetz AG, Vlisides PE (2023) Microstate analyses as an indicator of anesthesia-induced unconsciousness. Clin Neurophysiol 147:81–87. https://doi.org/10.1016/j.clinph.2023.01.007
Lehmann D, Faber PL, Galderisi S, Herrmann WM, Kinoshita T, Koukkou M, Mucci A, Pascual-Marqui RD, Saito N, Wackermann J, Winterer G, Koenig T (2005) EEG microstate duration and syntax in acute, medication-naïve, first-episode schizophrenia: a multi-center study. Psychiatry Res-Neuroimaging 138(2):141–156. https://doi.org/10.1016/j.pscychresns.2004.05.007
Lizier JT, Prokopenko M, Zomaya AY (2012) Local measures of information storage in complex distributed computation. Inf Sci 208:39–54. https://doi.org/10.1016/j.ins.2012.04.016
Lynn CW, Cornblath EJ, Papadopoulos L, Bertolero MA, Bassett DS (2021) Broken detailed balance and entropy production in the human brain. Proc Nat Acadamy Sci 118(47):e2109889118. https://doi.org/10.1073/pnas.2109889118
Michel CM, Koenig T (2018) EEG microstates as a tool for studying the temporal dynamics of whole-brain neuronal networks: A review. NeuroImage 180:577–593. https://doi.org/10.1016/j.neuroimage.2017.11.062
Milz P, Pascual-Marqui RD, Achermann P, Kochi K, Faber PL (2017) The EEG microstate topography is predominantly determined by intracortical sources in the alpha band. NeuroImage 162:353–361. https://doi.org/10.1016/J.NEUROIMAGE.2017.08.058
Murray MM, Brunet D, Michel CM (2008) Topographic ERP analyses: a step-by-step tutorial review. Brain Topogr 20(4):249–264. https://doi.org/10.1007/s10548-008-0054-5
Pascual-Marqui RD, Michel CM, Lehmann D (1995) Segmentation of brain electrical activity into microstates: model estimation and validation. IEEE Trans Biomed Eng 42(7):658–665. https://doi.org/10.1109/10.391164
Purdon PL, Pierce ET, Mukamel EA, Prerau MJ, Walsh JL, Wong KFK, Salazar-Gomez AF, Harrell PG, Sampson AL, Cimenser A, Ching S, Kopell NJ, Tavares-Stoeckel C, Habeeb K, Merhar R, Brown EN (2013) Electroencephalogram signatures of loss and recovery of consciousness from propofol. Proc Natl Acad Sci USA. https://doi.org/10.1073/pnas.1221180110
Sanz Perl Y, Bocaccio H, Pallavicini C, Pérez-Ipiña I, Laureys S, Laufs H, Kringelbach M, Deco G, Tagliazucchi E (2021) Nonequilibrium brain dynamics as a signature of consciousness. Phys Rev E 104(1):014411. https://doi.org/10.1103/PHYSREVE.104.014411/FIGURES/5/MEDIUM
Schiller B, Koenig T, Heinrichs M (2019) Oxytocin modulates the temporal dynamics of resting EEG networks. Sci Rep. https://doi.org/10.1038/s41598-019-49636-6
Schumacher J, Peraza LR, Firbank M, Thomas AJ, Kaiser M, Gallagher P, O’Brien JT, Blamire AM, Taylor JP (2019) Dysfunctional brain dynamics and their origin in Lewy body Dementia. Brain 142(6):1767–1782. https://doi.org/10.1093/BRAIN/AWZ069
Seabold S, Perktold J (2010) statsmodels: econometric and statistical modeling with python. In 9th python in science conference
Shi W, Li Y, Liu Z, Li J, Wang Q, Yan X, Wang G (2020) Non-canonical microstate becomes salient in high density EEG during propofol-induced altered states of consciousness. Int J Neural Syst. https://doi.org/10.1142/S0129065720500057
Strik WK, Dierks T, Becker T, Lehmann D (1995) Larger topographical variance and decreased duration of brain electric microstates in depression. J Neural Transm 99(1–3):213–222. https://doi.org/10.1007/BF01271480
Strik WK, Chiaramonti R, Muscas GC, Paganini M, Mueller TJ, Fallgatter AJ, Versari A, Zappoli R (1997) Decreased EEG microstate duration and anteriorisation of the brain electrical fields in mild and moderate Dementia of the Alzheimer type. Psychiatry Res: Neuroimaging 75(3):183–191. https://doi.org/10.1016/S0925-4927(97)00054-1
Tait L, Tamagnini F, Stothart G, Barvas E, Monaldini C, Frusciante R, Volpini M, Guttmann S, Coulthard E, Brown JT, Kazanina N, Goodfellow M (2020) EEG microstate complexity for aiding early diagnosis of Alzheimer’s disease. Sci Rep. https://doi.org/10.1038/s41598-020-74790-7
Tomescu MI, Rihs TA, Becker R, Britz J, Custo A, Grouiller F, Schneider M, Debbané M, Eliez S, Michel CM (2014) Deviant dynamics of EEG resting state pattern in 22q11.2 deletion syndrome adolescents: a vulnerability marker of schizophrenia? Schizophr Res 157(1–3):175–181. https://doi.org/10.1016/J.SCHRES.2014.05.036
Toplutaş E, Aydın F, Hanoğlu L (2023) EEG Microstate Analysis in patients with disorders of consciousness and its clinical significance. Brain Topogr 1:1–11. https://doi.org/10.1007/S10548-023-00939-Y/FIGURES/5
Van Rossum G, Drake FL (2009) Python 3 reference manual. CreateSpace 121:242
Virtanen P, Gommers R, Oliphant TE, Haberland M, Reddy T, Cournapeau D, Burovski E, Peterson P, Weckesser W, Bright J, van der Walt SJ, Brett M, Wilson J, Millman KJ, Mayorov N, Nelson ARJ, Jones E, Kern R, Larson E, …, Vázquez-Baeza Y (2020) SciPy 1.0: fundamental algorithms for scientific computing in Python. Nat Methods 17(3):261–272. https://doi.org/10.1038/s41592-019-0686-2
Von Wegner F (2018) Partial autoinformation to characterize symbolic sequences. Front Physiol 9(OCT):1–14. https://doi.org/10.3389/fphys.2018.01382
von Wegner F, Laufs H (2018) Information-theoretical analysis of EEG microstate sequences in python. Front Neuroinformatics 12(June):1–10. https://doi.org/10.3389/fninf.2018.00030
von Wegner F, Tagliazucchi E, Laufs H (2017) Information-theoretical analysis of resting state EEG microstate sequences - non-markovianity, non-stationarity and periodicities. NeuroImage. https://doi.org/10.1016/j.neuroimage.2017.06.062
von Wegner F, Knaut P, Laufs H (2018) EEG microstate sequences from different clustering algorithms are information-theoretically invariant. Front Comput Neurosci 12(August):1–14. https://doi.org/10.3389/fncom.2018.00070
von Wegner F, Bauer S, Rosenow F, Triesch J, Laufs H (2021) EEG microstate periodicity explained by rotating phase patterns of resting-state alpha oscillations. NeuroImage. https://doi.org/10.1016/j.neuroimage.2020.117372
Wibral M, Lizier JT, Vögler S, Priesemann V, Galuske R (2014) Local active information storage as a tool to understand distributed neural information processing. Front Neuroinformatics. https://doi.org/10.3389/fninf.2014.00001
Zanesco AP, King BG, Skwara AC, Saron CD (2020) Within and between-person correlates of the temporal dynamics of resting EEG microstates. NeuroImage. https://doi.org/10.1016/j.neuroimage.2020.116631
Zhang K, Shi W, Wang C, Li Y, Liu Z, Liu T, Li J, Yan X, Wang Q, Cao Z, Wang G (2021) Reliability of EEG microstate analysis at different electrode densities during propofol-induced transitions of brain states. NeuroImage 231:117861. https://doi.org/10.1016/J.NEUROIMAGE.2021.117861
Acknowledgements
This work was funded by the German Research Foundation (DFG), project number 440536202. The authors thank the researchers Chennu et al. (2016) for the publication of their dataset.
Author information
Authors and Affiliations
Contributions
Study concept and design: GH, FvW. Analyses and figures: GH, FvW, HL. All authors contributed to manuscript writing and reviewing. All authors read and approved the submitted version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Handling Editor: Samuel Brod.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hermann, G., Tödt, I., Tagliazucchi, E. et al. Propofol Reversibly Attenuates Short-Range Microstate Ordering and 20 Hz Microstate Oscillations. Brain Topogr 37, 329–342 (2024). https://doi.org/10.1007/s10548-023-01023-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10548-023-01023-1