Abstract
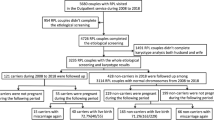
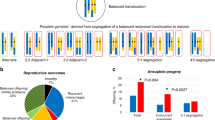
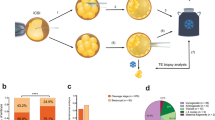
Balanced chromosomal translocation is one of chromosomal variations. Carriers of balanced chromosomal translocations have an increased risk of spontaneous miscarriage. To avoid the risk, preimplantation genetic testing (PGT) using comprehensive genomic copy number analysis has been developed. This study aimed to verify whether and how embryos from couples in which one partner is a balanced translocation carrier have a higher ratio of chromosomal abnormalities. A total of 894 biopsied trophectoderms (TEs) were obtained from 130 couples in which one partner was a balanced translocation carrier (Robertsonian translocation, reciprocal translocation, or intrachromosomal inversion) and grouped as PGT-SR. Conversely, 3269 TEs from 697 couples who experienced recurrent implantation failure or recurrent pregnancy loss were included in the PGT-A group. The transferable blastocyst ratio was significantly lower in the PGT-SR group, even when bias related to the sample number and patient age was corrected. Subgroup analysis of the PGT-SR group revealed that the transferable blastocyst ratio was higher in the Robertsonian translocation group. Because the PGT-SR group had a higher proportion of untransferable embryos than the PGT-A group, PGT using comprehensive genomic copy number analysis was more beneficial for balanced translocation carriers than for infertility patients without chromosomal translocations. The frequencies of de novo aneuploidies were further analyzed, and the frequency in the PGT-SR group was lower than that in the PGT-A group. Therefore, we could not confirm the existence of interchromosomal effects in this study.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Jacobs PA, Browne C, Gregson N, Joyce C, White H. Estimates of the frequency of chromosome abnormalities detectable in unselected newborns using moderate levels of banding. J Med Genet. 1992;29:103–8.
Sugiura-Ogasawara M, Ozaki Y, Sato T, Suzumori N, Suzumori K. Poor prognosis of recurrent aborters with either maternal or paternal reciprocal translocations. Fertil Steril. 2004;81:367–73.
Madan K, Nieuwint AW, van Bever Y. Recombination in a balanced complex translocation of a mother leading to a balanced reciprocal translocation in the child. Review of 60 cases of balanced complex translocations. Hum Genet. 1997;99:806–15.
Ogilvie CM, Braude P, Scriven PN. Successful pregnancy outcomes after preimplantation genetic diagnosis (PGD) for carriers of chromosome translocations. Hum Fertil (Camb). 2001;4:168–71.
Kato K, Aoyama N, Kawasaki N, Hayashi H, Xiaohui T, Abe T, et al. Reproductive outcomes following preimplantation genetic diagnosis using fluorescence in situ hybridization for 52 translocation carrier couples with a history of recurrent pregnancy loss. J Hum Genet. 2016;61:687–92.
Kurahashi H, Kato T, Miyazaki J, Nishizawa H, Nishio E, Furukawa H, et al. Preimplantation genetic diagnosis/screening by comprehensive molecular testing. Reprod Med Biol. 2016;15:13–19.
Tulay P, Gultomruk M, Findikli N, Yagmur E, Bahceci M. Is the interchromosomal effect present in embryos derived from Robertsonian and reciprocal translocation carriers particularly focusing on chromosome 10 rearrangements? Zygote 2015;23:908–15.
Bartels CB, Makhijani R, Godiwala P, Bartolucci A, Nulsen JC, Grow DR, et al. In vitro fertilization outcomes after preimplantation genetic testing for chromosomal structural rearrangements comparing fluorescence in-situ hybridization, microarray comparative genomic hybridization, and next-generation sequencing. F S Rep. 2020;1:249–56.
Nakano T, Ammae M, Satoh M, Mizuno S, Nakaoka Y, Morimoto Y. Analysis of clinical outcomes and meiotic segregation modes following preimplantation genetic testing for structural rearrangements using aCGH/NGS in couples with balanced chromosome rearrangement. Reprod Med Biol. 2022;21:e12476.
Viotti M. Preimplantation genetic testing for chromosomal abnormalities: aneuploidy, mosaicism, and structural rearrangements. Genes. 2020;11:602.
Bono S, Biricik A, Spizzichino L, Nuccitelli A, Minasi MG, Greco E, et al. Validation of a semiconductor next-generation sequencing-based protocol for preimplantation genetic diagnosis of reciprocal translocations. Prenat Diagn. 2015;35:938–44.
Iwasa T, Kuwahara A, Takeshita T, Taniguchi Y, Mikami M, Irahara M. Preimplantation genetic testing for aneuploidy and chromosomal structural rearrangement: a summary of a nationwide study by the Japan Society of Obstetrics and Gynecology. Reprod Med Biol. 2023;22:e12518.
Kato K, Kuroda T, Yamadera-Egawa R, Ezoe K, Aoyama N, Usami A, et al. Preimplantation genetic testing for aneuploidy for recurrent pregnancy loss and recurrent implantation failure in minimal ovarian stimulation cycle for women aged 35-42 years: live birth rate, developmental follow-up of children, and embryo ranking. Reprod Sci. 2023;30:974–83.
Takeuchi K. Pre-implantation genetic testing: past, present, future. Reprod Med Biol. 2021;20:27–40.
Sato T, Sugiura-Ogasawara M, Ozawa F, Yamamoto T, Kato T, Kurahashi H, et al. Preimplantation genetic testing for aneuploidy: a comparison of live birth rates in patients with recurrent pregnancy loss due to embryonic aneuploidy or recurrent implantation failure. Hum Reprod. 2019;34:2340–8.
Ma X, Xu X, Mao B, Liu H, Li H, Liu K, et al. Chromosomal analysis for embryos from balanced chromosomal rearrangement carriers using next generation sequencing. Mol Reprod Dev. 2021;88:362–70.
Shetty S, Nair J, Johnson J, Shetty N, Kumar AJ, Thondehalmath N, et al. Preimplantation genetic testing for couples with balanced chromosomal rearrangements. J Reprod Infertil. 2022;23:213–23.
Park SJ, Min JY, Kang JS, Yang BG, Hwang SY, Han SH. Chromosomal abnormalities of 19,000 couples with recurrent spontaneous abortions: a multicenter study. Fertil Steril. 2022;117:1015–25.
Ogur C, Kahraman S, Griffin DK, Cinar Yapan C, Tufekci MA, Cetinkaya M, et al. PGT for structural chromosomal rearrangements in 300 couples reveals specific risk factors but an interchromosomal effect is unlikely. Reprod Biomed Online. 2023;46:713–27.
Xie Y, Xu Y, Wang J, Miao B, Zeng Y, Ding C, et al. Preliminary analysis of numerical chromosome abnormalities in reciprocal and Robertsonian translocation preimplantation genetic diagnosis cases with 24-chromosomal analysis with an aCGH/SNP microarray. J Assist Reprod Genet. 2018;35:177–86.
Grande M, Borrell A, Garcia-Posada R, Borobio V, Muñoz M, Creus M, et al. The effect of maternal age on chromosomal anomaly rate and spectrum in recurrent miscarriage. Hum Reprod. 2012;27:3109–17.
Yuan P, Zheng L, Ou S, Zhao H, Li R, Luo H, et al. Evaluation of chromosomal abnormalities from preimplantation genetic testing to the reproductive outcomes: a comparison between three different structural rearrangements based on next-generation sequencing. J Assist Reprod Genet. 2021;38:709–18.
Estop AM, Cieply K, Munne S, Surti U, Wakim A, Feingold E. Is there an interchromosomal effect in reciprocal translocation carriers? Sperm FISH studies. Hum Genet. 2000;106:517–24.
Miller DE. The interchromosomal effect: different meanings for different organisms. Genetics. 2020;216:621–31.
Boynukalin FK, Gultomruk M, Turgut NE, Rubio C, Rodrigo L, Yarkiner Z, et al. The impact of patient, embryo, and translocation characteristics on the ploidy status of young couples undergoing preimplantation genetic testing for structural rearrangements (PGT-SR) by next generation sequencing (NGS). J Assist Reprod Genet. 2021;38:387–96.
Fan J, Zhang X, Chen Y, Zhang J, Zhang L, Bi X, et al. Exploration of the interchromosomal effects in preimplantation genetic testing for structural rearrangements based on next-generation sequencing. Mol Genet Genom Med. 2022;10:e2017.
Mayeur A, Ahdad N, Hesters L, Grynberg M, Romana S, Sonigo C, et al. Does the prognosis after PGT for structural rearrangement differ between female and male translocation carriers? Reprod Biomed Online. 2020;40:684–92.
Wang J, Li D, Xu Z, Diao Z, Zhou J, Lin F, et al. Analysis of meiotic segregation modes in biopsied blastocysts from preimplantation genetic testing cycles of reciprocal translocations. Mol Cytogenet. 2019;12:11.
Acknowledgements
We would like to thank all the subjects for participating in this study. We also thank Yumiko Ondo and Rino Abiru for technical assistance.
Author information
Authors and Affiliations
Contributions
TY designed the study. AY organized the study and drafted the manuscript. TK, NK, KK contributed to acquisition of data. KSY and YK analyzed the data. TI, AK, TT, YT, MM, MI reviewed the manuscript critically. All authors contributed to the data interpretation. All authors agree to be accountable for all aspects of the work and ensure that questions related to the accuracy or integrity of any part of the study are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yamazaki, A., Kuroda, T., Kawasaki, N. et al. Preimplantation genetic testing using comprehensive genomic copy number analysis is beneficial for balanced translocation carriers. J Hum Genet 69, 41–45 (2024). https://doi.org/10.1038/s10038-023-01202-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s10038-023-01202-9