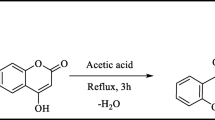
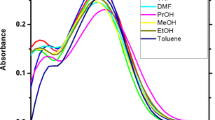
Abstract
The spectral properties of MBTC (4-((4-((Benzo[d]oxazol-2-ylthio)methyl)-1H-1,2,3-triazol-1-yl)methyl)-7-methoxy-2H-chromen-2-one), CBTC (4-((4(((5Chlorobenzo[d]oxazol-2-yl)thio)methyl)-1H-1,2,3-triazol-1-yl)methyl)-2H-benzo[h]chromen-2-one) and TBTC (4-((4-((Benzo[d]oxazol-2-ylthio)methyl)-1H-1,2,3-triazol-1-yl)methyl)6(tertbutyl)2Hchromen-2-one) were studied in series of solvents with different polarity at room temperature to explore their solvatochromic effect and dipole moment. Stokes shift revealed a bathochromic shift with varying solvent polarity for all molecules which implies π-π*transition. The ground state and excited state dipole moment of the molecules are calculated experimentally using salvatochromic methods like Lippert-Mataga, Bakhshiev, Kawaski-chamma-viallet, and Reichardt's microscopic solvent polarity functions and computationally by density functional theory (DFT) method. It is observed that the excited state dipole moment is higher than the ground state so synthesized molecules are more polar in the excited state than in the ground state. Using the DFT method HOMO and LUMO energy values were obtained and compared with values obtained by the cyclic voltammetry. Using the values of HOMO (highest occupied molecular orbital) and LUMO (lowest unoccupied molecular orbital) we have estimated energy gap, chemical hardness (ɳ), chemical softness (s), ionization potential (IP), electron affinity (EA), electronegativity (χ), electrophilicity (ω), and chemical potential (μ) of the molecules were estimated. The energy gap of MBTC, CBTC, and TBTC were found to be low, that is 3.861 eV, 3.822 eV, and 3.801 eV respectively, this indicates molecules are more reactive and it has the easiest π-π* transition. Further electrophilic and nucleophilic sites were figured out using molecular electrostatic potential (MESP) which is useful in photochemical reactions. Hence the quantum chemical calculation and spectroscopic properties of the molecules can give a better understanding of their use in an optoelectronic device.









Similar content being viewed by others
Data Availability
Not Applicable.
Code Availability
Not Applicable.
References
Drexhage KH (2005) Structure and properties of laser dyes. Top Appl Phys 1:155–200
Christie RM, Lui C-H (1999) Studies of fluorescent dyes: part 1. An investigation of the electronic spectral properties of substituted coumarins. Dyes Pigm 42:85–93
Chemla DS, Zyss J (1987) Non-linear optical properties of organic molecules and crystals. Academic Press, New York
Trenor SR, Shultz AR, Love BJ, Long TE (2004) Coumarins in Polymers: From Light Harvesting to Photo-Cross-Linkable Tissue Scaffolds. Chem Rev 104:3059–3077
Emami S, Dadashpour S (2015) Current developments of coumarin-based anti-cancer agents in medicinal chemistry. Eur J Med Chem 102:611–630
Kostova I, Bhatia S, Grigorov P, Balkansky S, Parmar VS, Prasad AK, Saso L (2011) Coumarins as antioxidants. Curr Med Chem 18:3929–3951
Kathiravan MK, Salake AB, Chothe AS, Dudhe PB, Watode RP, Mukta MS, Gadhwe S (2012) The biology and chemistry of antifungal agents: A review. Bioorg Med Chem 20:5678–5698
Grover J, Jachak SM (2015) Coumarins as privileged scaffold for anti-inflammatory drug development. RSC Adv 5(49):38892–38905
Szwaczko K (2022) Coumarins synthesis and transformation via C–H bond activation—A review. Inorganics 10(2):23
Liu Z-Q, Wei Yu, Liu Z-L (1999) Antioxidative and prooxidative effects of coumarin derivatives on free radical initiated and photosensitized peroxidation of human low-density lipoprotein. Chem Phys Lipid 103:125–135
Voutsadaki S, Tsikalas GK, Klontzas E, Froudakis GE, Katerinopoulos HE (2010) A ‘“turn-on”’ coumarin-based fluorescent sensor with high selectivity for mercury ions in aqueous media. Chem Commun 46:3292–3294
Feng S, Gao Q, Gao X, Yin J, Jiao Y (2019) Fluorescent sensor for copper(II) ions based on coumarin derivative and its application in cell imaging. Inorg Chem Commun 102:51–56
Pramod AG, Nadaf YF, Renuka CG (2019) Synthesis, photophysical, quantum chemical investigation, linear and non-linear optical properties of coumarin derivative: Optoelectronic and optical limiting application. Spectrochim Acta Part A Mol Biomol Spectrosc 223:117288
Moog RS, Kim DD, Oberle JJ, Ostrowski SG (2004) Solvent effects on electronic transitions of highly dipolar dyes: a comparison of three approaches. J Phys Chem A 108:9294–9301
Kaufman JJ, Predney R (1972) Extension of indo formalism to d orbitals and parameters for second-row atoms. Int J Quant Chem 231–242
Egan D, O’kennedy R, Mow E, Cox D, Prosser E, Thornes RD (1990) The pharmacology, metabolism, analysis and applications of compounds Coumarin and Coumarin4elated. Drug Metabol Rev 22(5):503–529
Deepa HR, Thipperudrappa J, Suresh Kumar HM (2013) Effect of solvents on the spectroscopic properties of LD-489 & LD-473: Estimation of ground and excited state dipole moments by solvatochromic shift method. Spectrochim Acta Part A Mol Biomol Spectrosc 108:288–294
Liu X, Cole JM, Low KS (2013) Solvent effects on the UV−vis absorption and emission of optoelectronic coumarins: a comparison of three empirical solvatochromic models. J Phys Chem C 117:14731–14741
Kumar S, Rao VC, Rastogi RC (2001) Excited-state dipole moments of some hydroxycoumarin dyes using an efficient solvatochromic method based on the solvent polarity parameter. ETN, Spectrochimica Acta Part A 57:41–47
Giri R (1992) Temperature effect study upon the fluorescence emission of substituted coumarins. Specrrochimico Acta 48A:843–848
Moriya T (1984) Excited state reaction coumarins in aqueous solutions. II. The fluorescence quenching of 7-ethoxycoumarins by halide ions. Bull Chem Soc Jpn 57:1723–1730
Muhammad S, Abdullah G, Al-Sehemi AG, Pannipara M, Irfan A (2018) Design, characterization and nonlinear optical properties of coumarin appended chalcones: Use of a dual approach. Optik 164:5–15
De Haas MP, Warman JM (1952) Photon-induced moeecdl~ charge separation studled by nanosecond time-resolved microwave conductivity. Chem Phys 73:35–53
Lombardi JR (1969) Dipole moments of the lowest singlet π*←π states in phenol and aniline by the optical stark effect. J Chem Phys 50:3780
Rosell FI, Boxer SG (2003) Polarized absorption spectra of green fluorescent protein single crystals: Transition dipole moment directions. Biochemistry 42:177–183
Liptay W (1974) E. C. Lim (Eds.) Excited-state, Academic Press, New York
Kawski A, Kukliński B, Bojarski P (2005) Dipole moment of aniline in the excited S1 state from the thermochromic effect on electronic spectra. Chem Phys Lett 415:251–255
Kadam MML, Patil DS, Sekar N (2018) Red emitting coumarin based 4, 6-disubstituted-3-cyano-2-pyridones dyes – Synthesis, solvatochromism, linear and non-linear optical properties. J Mol Liq 11:164
Khatir–Hamdi N, Makhloufi-Chebli M, Grib H, Brahimi M, Silva AM (2018) Synthesis, DFT/TD-DFT theoretical studies and experimental solvatochromic shift methods on determination of ground and excited-state dipole moments of 3- (2-hydroxybenzoyl)coumarins. J Mol Struct 08:039
Cave RJ, Castner EW (2002) Time-dependent density functional theory investigation of the ground and excited states of coumarins 102, 152, 153, and 343. J Phys Chem A 106:12117–12123
Patil SK, Wari MN, Yohannan Panicker C, Inamdar SR (2014) Determination of ground and excited-state dipole moments of dipolar laser dyes by solvatochromic shift method. Spectrochim Acta Part A Mol Biomol Spectrosc 123:117–126
Shashikant Walki GH, Malimathb KMM, Naik S, Sutar SM, Savanur H, Naik L (2021) Synthesis, spectroscopic properties, and DFT correlative studies of 3,3 - carbonyl biscoumarin derivatives. J Mol Struct 1243:130781
Warde U, Sekar N (2017) Solvatochromic benzo[h] coumarins: Synthesis, solvatochromism. NLO, and DFT study, /Optical Materials 72:346–358
Paczkowski IM, Coelho FL, Campo LF (2020) 2,1,3-Benzothiadiazole dyes conjugated with benzothiazole and benzoxazole: Synthesis, Solvatochromism, and solid-state properties. J Mol Liq 319:114277
Nesaragi AR, Kamble RR, Bayannavar PK, Metre TV, Kariduraganavar MY, Margankop SB, Joshi SD, Kumbar VM (2021) Microwave facilitated one-pot three component synthesis of coumarin-benzoxazole clubbed 1,2,3- triazoles: Antimicrobial evaluation, molecular docking and in silico ADME studies. Synth Commun 51:3460–3472
Von E LIPPERT, pek$re & opiSohe b&hnmmg dee Dipole moments ammatiseber Verbindqen im ersten amregten SingdeMzmstand). 61:8
Bakhshiev G (1964) Opt Spectrosc (USSR) 16:821
Kawski A (2002) On the estimation of excited-state dipole moments from solvatochromic shifts of absorption and fluorescence spectra. Z Naturforsch A 57:255–262
Reichardt C (1594) Solvatochromic dyes as solvent polarity indicators. Chem Rev 94:2319–2358
Reichardt C (1988) Solvents and solvent effects in organic chemistry. Chemistry 11(1):105–113
Ravi M, Soujanya T, Samanta A, Radhakrishnan TP (1995) Excited-state dipole moments of some coumarin dyes from a solvatochromic method using the solvent polarity parameter, ETN. J Chem Soc Faraday Trans 91(17): 2739–2742
Patil MK, Kotresh MG, Inamdar SR (2019) A combined solvatochromic shift and TDDFT study probing solute-solvent interactions of blue fluorescent Alexa Fluor 350 dye: Evaluation of ground and excited-state dipole moments. Spectrochim Acta Part A Mol Biomol Spectrosc 215:142–152
Geethanjali HS, Nagaraja D, Melavanki RM (2015) Estimation of Dipole Moments and Quantum Yield of 5-chloro-2-methoxyphenyl Boronic Acid in Different Solvents Environment. J Fluoresc 25:745–753
Gayathri R, Arivazhagan M (2011) Vibrational spectroscopy investigation and HOMO, LUMO analysis using DFT (B3LYP) on the structure of 1,3-dichloro 5-nitrobenzene. Spectrochim Acta Part A 81:242–250
Naik L, Maridevarmath CV, Khazi IAM, Malimath GH (2018) Photophysical and computational studies on optoelectronically active thiophene substituted 1,3,4-oxadiazole derivatives. J Photochem 09:038
Kumari R, Varghese A, George L, Sudhakar YN (2017) Effect of solvent polarity on the photophysical properties of chalcone derivatives. RSC Adv 7:24204–24214
Patil S, Desai VR, Basanagouda M, Hunagund SM, Tuwar S, Sidarai AH (2013) Estimation of photophysical and electrochemical parameters of bioactive thiadiazole derivative. J Fluoresc
Akshaya KB, Varghese A, Sudhakar YN, Lobo PL, George L (2017) Study on photophysical properties of n-arylphthalamic acid derivative containing 1, 2, 4-triazole scaffold. J Fluoresc 27:1909–1922
Leonat L, Sbârcea G, Brânzoi IV (2013) Cyclic voltammetry for energy levels estimation of organic materials. UPB Sci Bull Ser B 75:3
Strojnik M, Omerzu A, Majki A, Mihailovic PM, Lukan J, Bavdek G, Bratina G, Cvetko D, Topolovsek P, Mihailovic D (2011) Ionization energy and energy gap structure of MoSI molecular wires: Kelvin Probe, ultraviolet photoelectron spectroscopy, and cyclic voltammetry measurements. Langmuir 27:4296–4299
Acknowledgements
The authors are thankful to the authorities of USIC, KUD, for providing the instrumental facility for this research work.
Author information
Authors and Affiliations
Contributions
All authors contributed to this study conceptions and design. Sample Preparation, data collection and analysis were Performed by Bi Bi Ayisha Mulla, Material synthesized by Dr. Aravind R Nesargi and Prof. Ravindra R Kamble. the first draft of the manuscript was written by Bi Bi Ayisha Mulla. Computational work has done by Dr. Mussuvir Pasha K M. Draft editing and overall review done by Dr. Malatesh S Pujar and Prof. Ashok H Sidarai. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Not Applicable.
Consent to Participate
Not Applicable.
Consent for Publication
Not Applicable.
Conflict of Interest
The authors declare they have no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mulla, B.B.A., Nesaragi, A.R., M, M.P.K. et al. Experimental and Theoretical Spectroscopic Investigation on Coumarin Based Derivatives for Non-Linear Optoelectronics Application. J Fluoresc 33, 161–175 (2023). https://doi.org/10.1007/s10895-022-03046-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-022-03046-6