Abstract
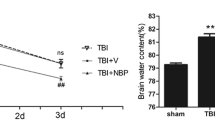
Oxidative stress and neuroinflammation are deemed the prime causes of neurological damage after traumatic brain injury (TBI). Catalpol, an active ingredient of Rehmannia glutinosa, has been suggested to possess antioxidant and anti-inflammatory properties. This study was designed to investigate the protective effects of catalpol against TBI and the underlying mechanisms of action of catalpol. A rat model of TBI was induced by controlled cortical impact. Catalpol (10 mg/kg) or vehicle was administered via intravenous injection 1 h post trauma and then once daily for 3 consecutive days. Following behavioural tests performed 72 h after TBI, the animals were sacrificed and pericontusional areas of the brain were collected for neuropathological experiments and analysis. Treatment with catalpol significantly ameliorated neurological impairment, blood–brain barrier disruption, cerebral oedema, and neuronal apoptosis after TBI (P < 0.05). Catalpol also attenuated TBI-induced oxidative insults, as evidenced by reduced reactive oxygen species generation; decreased malondialdehyde levels; and enhanced superoxide dismutase, catalase and glutathione peroxidase activity (P < 0.05). Catalpol promoted the nuclear translocation of nuclear factor erythroid 2-related factor 2 and the expression of its downstream antioxidant enzyme HO-1 following TBI (P < 0.05). Moreover, catalpol treatment markedly inhibited posttraumatic microglial activation and neutrophil infiltration, suppressed NLRP3 inflammasome activation and reduced the production of the proinflammatory cytokine IL-1β (P < 0.05). Taken together, these findings reveal that catalpol provides neuroprotection against oxidative stress and neuroinflammation after TBI in rats. Therefore, catalpol may be a novel treatment strategy for TBI patients.







Similar content being viewed by others
Data Availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Capizzi A, Woo J, Verduzco-Gutierrez M (2020) Traumatic brain injury: an overview of epidemiology, pathophysiology, and medical management. Med Clin 104:213–238. https://doi.org/10.1016/j.mcna.2019.11.001
Dixon KJ (2017) Pathophysiology of traumatic brain injury. Phys Med Rehabil Clin N Am 28:215–225. https://doi.org/10.1016/j.pmr.2016.12.001
Kolias AG, Rubiano AM, Figaji A, Servadei F, Hutchinson PJ (2019) Traumatic brain injury: global collaboration for a global challenge. Lancet Neurol 18:136–137. https://doi.org/10.1016/S1474-4422(18)30494-0
Khatri N, Thakur M, Pareek V, Kumar S, Sharma S, Datusalia AK (2018) Oxidative stress: major threat in traumatic brain injury. CNS Neurol Disord Drug Targets 17:689–695. https://doi.org/10.2174/1871527317666180627120501
Simon DW, McGeachy MJ, Bayır H, Clark RS, Loane DJ, Kochanek PM (2017) The far-reaching scope of neuroinflammation after traumatic brain injury. Nat Rev Neurol 13:171–191. https://doi.org/10.1038/nrneurol.2017.13
Abdul-Muneer P, Chandra N, Haorah J (2015) Interactions of oxidative stress and neurovascular inflammation in the pathogenesis of traumatic brain injury. Mol Neurobiol 51:966–979. https://doi.org/10.1007/s12035-014-8752-3
Zhang L, Wang H (2018) Targeting the NF-E2-related factor 2 pathway: a novel strategy for traumatic brain injury. Mol Neurobiol 55:1773–1785. https://doi.org/10.1007/s12035-017-0456-z
Ma Q (2013) Role of nrf2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol 53:401–426. https://doi.org/10.1146/annurev-pharmtox-011112-140320
O’Brien WT, Pham L, Symons GF, Monif M, Shultz SR, McDonald SJ (2020) The NLRP3 inflammasome in traumatic brain injury: potential as a biomarker and therapeutic target. J Neuroinflammation 17:1–12. https://doi.org/10.1186/s12974-020-01778-5
Tschopp J, Schroder K (2010) NLRP3 inflammasome activation: the convergence of multiple signalling pathways on ROS production? Nat Rev Immunol 10:210–215. https://doi.org/10.1038/nri2725
Dominic A, Le N-T, Takahashi M (2022) Loop between NLRP3 inflammasome and reactive oxygen species. Antioxid Redox Signal 36:784–796. https://doi.org/10.1089/ars.2020.8257
Jiang B, Shen R, Bi J, Tian X, Hinchliffe T, Xia Y (2015) Catalpol: a potential therapeutic for neurodegenerative diseases. Curr Med Chem 22:1278–1291. https://doi.org/10.2174/0929867322666150114151720
Wang Q, Xing M, Chen W, Zhang J, Qi H, Xu X (2012) HPLC–APCI–MS/MS method for the determination of catalpol in rat plasma and cerebrospinal fluid: application to an in vivo pharmacokinetic study. J Pharm Biomed Anal 70:337–343. https://doi.org/10.1016/j.jpba.2012.05.016
Wang LY, Yu X, Li XX, Zhao YN, Wang CY, Wang ZY, He ZY (2019) Catalpol exerts a neuroprotective effect in the MPTP mouse model of Parkinson’s disease. Front Aging Neurosci 11:316. https://doi.org/10.3389/fnagi.2019.00316
Huang JZ, Wu J, Xiang S, Sheng S, Jiang Y, Yang Z, Hua F (2016) Catalpol preserves neural function and attenuates the pathology of Alzheimer’s disease in mice. Mol Med Rep 13:491–496. https://doi.org/10.3892/mmr.2015.4496
Wang YL, Wu HR, Zhang SS, Xiao HL, Yu J, Ma YY, Zhang YD, Liu Q (2021) Catalpol ameliorates depressive-like behaviors in CUMS mice via oxidative stress-mediated NLRP3 inflammasome and neuroinflammation. Transl Psychiatry 11:353. https://doi.org/10.1038/s41398-021-01468-7
Zheng XW, Yang WT, Chen S, Xu QQ, Shan CS, Zheng GQ, Ruan JC (2017) Neuroprotection of catalpol for experimental acute focal ischemic stroke: Preclinical evidence and possible mechanisms of antioxidation, anti-inflammation, and antiapoptosis. Oxid Med Cell Longev 2017:5058609. https://doi.org/10.1155/2017/5058609
Dixon CE, Clifton GL, Lighthall JW, Yaghmai AA, Hayes RL (1991) A controlled cortical impact model of traumatic brain injury in the rat. J Neurosci Methods 39:253–262. https://doi.org/10.1016/0165-0270(91)90104-8
Wang H, Xu X, Yin Y, Yu S, Ren H, Xue Q, Xu X (2020) Catalpol protects vascular structure and promotes angiogenesis in cerebral ischemic rats by targeting HIF-1alpha/VEGF. Phytomedicine 78:153300. https://doi.org/10.1016/j.phymed.2020.153300
Xu X, Yin D, Ren H, Gao W, Li F, Sun D, Wu Y, Zhou S, Lyu L, Yang M, Xiong J, Han L, Jiang R, Zhang J (2018) Selective NLRP3 inflammasome inhibitor reduces neuroinflammation and improves long-term neurological outcomes in a murine model of traumatic brain injury. Neurobiol Dis 117:15–27. https://doi.org/10.1016/j.nbd.2018.05.016
Ni H, Rui Q, Xu Y, Zhu J, Gao F, Dang B, Li D, Gao R, Chen G (2018) RACK1 upregulation induces neuroprotection by activating the IRE1-XBP1 signaling pathway following traumatic brain injury in rats. Exp Neurol 304:102–113. https://doi.org/10.1016/j.expneurol.2018.03.003
Rui Q, Ni H, Gao F, Dang B, Li D, Gao R, Chen G (2018) LRRK2 contributes to secondary brain injury through a p38/Drosha signaling pathway after traumatic brain injury in rats. Front Cell Neurosci 12:51. https://doi.org/10.3389/fncel.2018.00051
Rui Q, Ni H, Lin X, Zhu X, Li D, Liu H, Chen G (2019) Astrocyte-derived fatty acid-binding protein 7 protects blood-brain barrier integrity through a caveolin-1/MMP signaling pathway following traumatic brain injury. Exp Neurol 322:113044. https://doi.org/10.1016/j.expneurol.2019.113044
Ni H, Rui Q, Lin X, Li D, Liu H, Chen G (2019) 2-BFI provides neuroprotection against inflammation and necroptosis in a rat model of traumatic brain injury. Front Neurosci 13:674. https://doi.org/10.3389/fnins.2019.00674
Kaspar JW, Niture SK, Jaiswal AK (2009) Nrf 2: INrf2 (Keap1) signaling in oxidative stress. Free Radic Biol Med 47:1304–1309. https://doi.org/10.1016/j.freeradbiomed.2013.02.008
Galgano M, Toshkezi G, Qiu X, Russell T, Chin L, Zhao L-R (2017) Traumatic brain injury: current treatment strategies and future endeavors. Cell Transplant 26:1118–1130. https://doi.org/10.1177/0963689717714102
Yang C, Shi Z, You L, Du Y, Ni J, Yan D (2020) Neuroprotective effect of catalpol via anti-oxidative, anti-inflammatory, and anti-apoptotic mechanisms. Front Pharmacol 11:690. https://doi.org/10.3389/fphar.2020.00690
Ng SY, Lee AYW (2019) Traumatic brain injuries: pathophysiology and potential therapeutic targets. Front Cell Neurosci 13:528. https://doi.org/10.3389/fncel.2019.00528
Cornelius C, Crupi R, Calabrese V, Graziano A, Milone P, Pennisi G, Radak Z, Calabrese EJ, Cuzzocrea S (2013) Traumatic brain injury: oxidative stress and neuroprotection. Antioxid Redox Signal 19:836–853. https://doi.org/10.1089/ars.2012.4981
Rodriguez-Rodriguez A, Jose Egea-Guerrero J, Murillo-Cabezas F, Carrillo-Vico A (2014) Oxidative stress in traumatic brain injury. Curr Med Chem 21:1201–1211. https://doi.org/10.2174/0929867321666131217153310
Di Pietro V, Yakoub KM, Caruso G, Lazzarino G, Signoretti S, Barbey AK, Tavazzi B, Lazzarino G, Belli A, Amorini AM (2020) Antioxidant therapies in traumatic brain injury. Antioxidants 9:260. https://doi.org/10.1016/j.pmr.2016.12.001
Xia H, Wang D, Guo X, Wu K, Huang F, Feng Y (2020) Catalpol protects against spinal cord injury in mice through regulating microRNA-142-mediated HMGB1/TLR4/NF-kappaB signaling pathway. Front Pharmacol 11:630222. https://doi.org/10.3389/fphar.2020.630222
Bellezza I, Giambanco I, Minelli A, Donato R (2018) Nrf2-Keap1 signaling in oxidative and reductive stress. Biochim Biophys Acta Mol Cell Res 1865:721–733. https://doi.org/10.1016/j.bbamcr.2018.02.010
Yan W, Wang H-D, Hu Z-G, Wang Q-F, Yin H-X (2008) Activation of Nrf2–ARE pathway in brain after traumatic brain injury. Neurosci Lett 431:150–154. https://doi.org/10.1016/j.neulet.2007.11.060
Thapa K, Khan H, Singh TG, Kaur A (2021) Traumatic brain injury: mechanistic insight on pathophysiology and potential therapeutic targets. J Mol Neurosci 71:1725–1742. https://doi.org/10.1007/s12031-021-01841-7
Jin W, Wang H, Yan W, Zhu L, Hu Z, Ding Y, Tang K (2009) Role of Nrf2 in protection against traumatic brain injury in mice. J Neurotrauma 26:131–139. https://doi.org/10.1089/neu.2008.0655
Corps KN, Roth TL, McGavern DB (2015) Inflammation and neuroprotection in traumatic brain injury. JAMA Neurol 72:355–362. https://doi.org/10.1001/jamaneurol.2014.3558
Ramlackhansingh AF, Brooks DJ, Greenwood RJ, Bose SK, Turkheimer FE, Kinnunen KM, Gentleman S, Heckemann RA, Gunanayagam K, Gelosa G (2011) Inflammation after trauma: microglial activation and traumatic brain injury. Ann Neurol 70:374–383. https://doi.org/10.1002/ana.22455
Kamm K, VanderKolk W, Lawrence C, Jonker M, Davis AT (2006) The effect of traumatic brain injury upon the concentration and expression of interleukin-1β and interleukin-10 in the rat. J Trauma 60:152–157. https://doi.org/10.1097/01.ta.0000196345.81169.a1
Rasouli J, Lekhraj R, White NM, Flamm ES, Pilla AA, Strauch B, Casper D (2012) Attenuation of interleukin-1beta by pulsed electromagnetic fields after traumatic brain injury. Neurosci Lett 519:4–8. https://doi.org/10.1016/j.neulet.2012.03.089
Xu X, Yin D, Ren H, Gao W, Li F, Sun D, Wu Y, Zhou S, Lyu L, Yang M (2018) Selective NLRP3 inflammasome inhibitor reduces neuroinflammation and improves long-term neurological outcomes in a murine model of traumatic brain injury. Neurobiol Dis 117:15–27. https://doi.org/10.1016/j.nbd.2018.05.016
Irrera N, Pizzino G, Calò M, Pallio G, Mannino F, Famà F, Arcoraci V, Fodale V, David A, Francesca C (2017) Lack of the Nlrp3 inflammasome improves mice recovery following traumatic brain injury. Front Pharmacol 8:459. https://doi.org/10.3389/fphar.2017.00459
Clausen F, Hånell A, Björk M, Hillered L, Mir AK, Gram H, Marklund N (2009) Neutralization of interleukin-1β modifies the inflammatory response and improves histological and cognitive outcome following traumatic brain injury in mice. Eur J Neurosci 30:385–396. https://doi.org/10.1111/j.1460-9568.2009.06820
Irrera N, Russo M, Pallio G, Bitto A, Mannino F, Minutoli L, Altavilla D, Squadrito F (2020) The role of NLRP3 inflammasome in the pathogenesis of traumatic brain injury. Int J Mol Sci 21:6204. https://doi.org/10.3390/ijms21176204
Hennig P, Garstkiewicz M, Grossi S, Di Filippo M, French LE, Beer HD (2018) The crosstalk between Nrf2 and inflammasomes. Int J Mol Sci. https://doi.org/10.3390/ijms19020562
Ikram M, Park HY, Ali T, Kim MO (2021) Melatonin as a potential regulator of oxidative stress, and neuroinflammation: mechanisms and implications for the management of brain injury-induced neurodegeneration. J Inflamm Res 14:6251. https://doi.org/10.2147/JIR.S334423
Zhao XJ, Zhu HY, Wang XL, Lu XW, Pan CL, Xu L, Liu X, Xu N, Zhang ZY (2022) Oridonin ameliorates traumatic brain injury-induced neurological damage by improving mitochondrial function and antioxidant capacity and suppressing neuroinflammation through the Nrf2 pathway. J Neurotrauma 39:530–543. https://doi.org/10.1089/neu.2021.0466
Yu J, Wang WN, Matei N, Li X, Pang JW, Mo J, Chen SP, Tang JP, Yan M, Zhang JH (2020) Ezetimibe attenuates oxidative stress and neuroinflammation via the AMPK/Nrf2/TXNIP pathway after MCAO in rats. Oxid Med Cell Longev 2020:4717258. https://doi.org/10.1155/2020/4717258
Hou Y, Wang Y, He Q, Li L, Xie H, Zhao Y, Zhao J (2018) Nrf2 inhibits NLRP3 inflammasome activation through regulating Trx1/TXNIP complex in cerebral ischemia reperfusion injury. Behav Brain Res 336:32–39. https://doi.org/10.1016/j.bbr.2017.06.027
Sun Y, Ji J, Zha Z, Zhao H, Xue B, Jin L, Wang L (2021) Effect and mechanism of catalpol on remyelination via regulation of the NOTCH1 signaling pathway. Front Pharmacol 12:628209. https://doi.org/10.3389/fphar.2021.628209
Wang J, Wan D, Wan G, Wang J, Zhang J, Zhu H (2019) Catalpol induces cell activity to promote axonal regeneration via the PI3K/AKT/mTOR pathway in vivo and in vitro stroke model. Ann Transl Med. https://doi.org/10.21037/atm.2019.11.101
Liu Z, Zhu P, Zhang L, Xiong B, Tao J, Guan W, Li C, Chen C, Gu J, Duanmu J, Zhang W (2018) Autophagy inhibition attenuates the induction of anti-inflammatory effect of catalpol in liver fibrosis. Biomed Pharmacother 103:1262–1271. https://doi.org/10.1016/j.biopha.2018.04.156
Acknowledgements
The authors thank American Journal Experts (https://www.aje.cn) for language editing of the manuscript.
Funding
This study was supported by the National Natural Science Foundation of China (Grant Nos. 81901254 and 82201538), the Project of Suzhou Health Talent Training (Grant No. GSWS2020105), the Project of Zhangjiagang Health Talent Training (Grant No. ZJGWSRC202004), the Project of Zhangjiagang Science and Technology (Grant No. ZKS2017), Six Talent Peaks in Jiangsu Province (SWYY-088, 2018 to B.L. Zhang), the 333 Project of Jiangsu Province (2022 to B.L. Zhang), and the Qing Lan Project in Jiangsu Province (2017 to B.L. Zhang).
Author information
Authors and Affiliations
Contributions
BZ and LZ designed the study. HN, QR, XK, and RG performed the experiments. BZ, LZ, HN, and QR performed the data analysis and interpretation. HN wrote the manuscript. BZ, LZ, and RG reviewed and revised the manuscript for final approval. All authors discussed the results and commented on the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical Appoval
The animal study was reviewed and approved by the Soochow University Institutional Animal Care and Use Committee (NO. ZJGYYLL-2021–09-008; Date: 7 Sep., 2021).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ni, H., Rui, Q., Kan, X. et al. Catalpol Ameliorates Oxidative Stress and Neuroinflammation after Traumatic Brain Injury in Rats. Neurochem Res 48, 681–695 (2023). https://doi.org/10.1007/s11064-022-03796-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-022-03796-6