Abstract
Purpose
Uvulopalatopharyngoplasty (UPPP) can aggravate lung inflammatory reactions in patients with obstructive sleep apnoea syndrome (OSAS). Dexmedetomidine (Dex) is a selective α-2 adrenoreceptor agonist that can alleviate lung injury. This study was designed to investigate the effects of Dex on oxygenation and inflammatory factors in patients undergoing UPPP in the early perioperative period.
Methods
Patients with OSAS undergoing UPPP were randomly allocated to the Dex Group or Control Group. Arterial blood gas analyses were performed, and the respiratory index (RI) and oxygenation index (OI) were calculated upon entering the operating room (T0) and immediately after surgery (T3). The inflammatory factors tumour necrosis factor-α (TNF-α), interleukin-6 (IL-6), and interleukin-10 (IL-10) were measured at T0 and T3.
Results
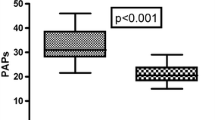
A total of 44 patients with OSAS were randomized. There was no significant difference in basic patient characteristics between the two groups. The preoperative RI and OI were not significantly different between the two groups, but they were altered immediately after surgery relative to the corresponding preoperative value (p < 0.05). Compared with the Control Group, the RI was significantly lower at T3 in the Dex Group (p < 0.001). However, there was no significant difference in the OI between the two groups (p = 0.128). The inflammatory factors TNF-α (p < 0.001) and IL-6 (p = 0.018) were lower, while IL-10 was higher in the Dex Group than in the Control Group (p < 0.001).
Conclusion
Dexmedetomidine can improve the oxygenation and inhibit the inflammatory response in patients undergoing UPPP in the early perioperative period.
Trial registration
The present clinical study has been registered at Clinical Trials under number NCT03612440.


Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Senaratna CV, Perret JL, Lodge CJ, Lowe AJ, Campbell BE, Matheson MC, Hamilton GS, Dharmage SC (2017) Prevalence of obstructive sleep apnea in the general population: a systematic review. Sleep Med Rev 34:70–81. https://doi.org/10.1016/j.smrv.2016.07.002
Ye L, Pien GW, Ratcliffe SJ, Björnsdottir E, Arnardottir ES, Pack AI, Benediktsdottir B, Gislason T (2014) The different clinical faces of obstructive sleep apnoea: a cluster analysis. Eur Respir J 44(6):1600–1607. https://doi.org/10.1183/09031936.00032314
Yeghiazarians Y, Jneid H, Tietjens JR, Redline S, Brown DL, El-Sherif N, Mehra R, Bozkurt B, Ndumele CE, Somers VK (2021) Obstructive sleep apnea and cardiovascular disease: a scientific statement from the American Heart Association. Circulation 144(3):e56–e67. https://doi.org/10.1161/CIR.0000000000000988
Sun WL, Wang JL, Jia GH, Mi WJ, Liao YX, Huang YW, Hu Z, Zhang LQ, Chen YH (2019) Impact of obstructive sleep apnea on pulmonary hypertension in patients with chronic obstructive pulmonary disease. Chin Med J (Engl) 132(11):1272–1282. https://doi.org/10.1097/CM9.0000000000000247
Labarca G, Gower J, Lamperti L, Dreyse J, Jorquera J (2020) Chronic intermittent hypoxia in obstructive sleep apnea: a narrative review from pathophysiological pathways to a precision clinical approach. Sleep Breath 24(2):751–760. https://doi.org/10.1007/s11325-019-01967-4
Emilsson ÖI, Sundbom F, Ljunggren M, Benediktsdottir B, Garcia-Aymerich J, Bui DS, Jarvis D, Olin AC, Franklin KA, Demoly P, Lindberg E, Janson C, Aspelund T, Gislason T (2021) Association between lung function decline and obstructive sleep apnoea: the ALEC study. Sleep Breath 25(2):587–596. https://doi.org/10.1007/s11325-020-02086-1
Abdeyrim A, Zhang Y, Li N, Zhao M, Wang Y, Yao X, Keyoumu Y, Yin T (2015) Impact of obstructive sleep apnea on lung volumes and mechanical properties of the respiratory system in overweight and obese individuals. BMC Pulm Med 15:76. https://doi.org/10.1186/s12890-015-0063-6
Sommer UJ, Heiser C, Gahleitner C, Herr RM, Hörmann K, Maurer JT, Stuck BA (2016) Tonsillectomy with uvulopalatopharyngoplasty in obstructive sleep apnea. Dtsch Arztebl Int 113(1–02):1–8. https://doi.org/10.3238/arztebl.2016.0001
Desborough JP (2000) The stress response to trauma and surgery. Br J Anaesth 85(1):109–117. https://doi.org/10.1093/bja/85.1.109
Mokra D, Kosutova P (2014) Biomarkers in acute lung injury. Respir Physiol Neurobiol 209:52–58. https://doi.org/10.1016/j.resp.2014.10.006
Li HD, Zhang QX, Mao Z, Xu XJ, Li NY, Zhang H (2015) Exogenous interleukin-10 attenuates hyperoxia-induced acute lung injury in mice. Exp Physiol 100(3):331–340. https://doi.org/10.1113/expphysiol.2014.083337
Shen Q, Xu G, Liu J, Wang L, Zhou Y, Yu Y, Lv C, Liu X (2020) Dexmedetomidine alleviates non-ventilation associated lung injury via modulating immunology phenotypes of macrophages. Life Sci 259:118249. https://doi.org/10.1016/j.lfs.2020.118249
Lee SH, Kim N, Lee CY, Ban MG, Oh YJ (2016) Effects of dexmedetomidine on oxygenation and lung mechanics in patients with moderate chronic obstructive pulmonary disease undergoing lung cancer surgery: a randomised double-blinded trial. Eur J Anaesthesiol 33(4):275–282. https://doi.org/10.1097/EJA.0000000000000405
Cui J, Gao M, Huang H, Huang X, Zeng Q (2020) Dexmedetomidine improves lung function by promoting inflammation resolution in patients undergoing totally thoracoscopic cardiac surgery. Oxid Med Cell Longev 2020:8638301. https://doi.org/10.1155/2020/8638301
Liu Z, Wang Y, Wang Y, Ning Q, Zhang Y, Gong C, Zhao W, Jing G, Wang Q (2016) Dexmedetomidine attenuates inflammatory reaction in the lung tissues of septic mice by activating cholinergic anti-inflammatory pathway. Int Immunopharmacol 35:210–216. https://doi.org/10.1016/j.intimp.2016.04.003
Sundar KM, Prchal JT (2017) The Cornerstone of the aberrant pathophysiology of obstructive sleep apnea: tissue responses to chronic sustained versus intermittent hypoxia. Am J Respir Cell Mol Biol 56(4):419–420. https://doi.org/10.1165/rcmb.2017-0028ED
Kimura H, Ota H, Kimura Y, Takasawa S (2019) Effects of intermittent hypoxia on pulmonary vascular and systemic diseases. Int J Environ Res Public Health 16(17):3101. https://doi.org/10.3390/ijerph16173101
Shimoda LA (2020) Cellular pathways promoting pulmonary vascular remodeling by hypoxia. Physiology (Bethesda) 35(4):222–233. https://doi.org/10.1152/physiol.00039.2019
Bao N, Tang B (2020) Organ-protective effects and the underlying mechanism of dexmedetomidine. Mediators Inflamm 2020:6136105. https://doi.org/10.1155/2020/6136105
Shi J, Yu T, Song K, Du S, He S, Hu X, Li X, Li H, Dong S, Zhang Y, Xie Z, Li C, Yu J (2021) Dexmedetomidine ameliorates endotoxin-induced acute lung injury in vivo and in vitro by preserving mitochondrial dynamic equilibrium through the HIF-1a/HO-1 signaling pathway. Redox Biol 41:101954. https://doi.org/10.1016/j.redox.2021.101954
Wu CY, Lu YF, Wang ML, Chen JS, Hsu YC, Yang FS, Cheng YJ (2018) Effects of dexmedetomidine infusion on inflammatory responses and injury of lung tidal volume changes during one-lung ventilation in thoracoscopic surgery: a randomized controlled trial. Mediators Inflamm 2018:2575910. https://doi.org/10.1155/2018/2575910
Yamakage M, Iwasaki S, Satoh JI, Namiki A (2008) Inhibitory effects of the alpha-2 adrenergic agonists clonidine and dexmedetomidine on enhanced airway tone in ovalbumin-sensitized guinea pigs. Eur J Anaesthesiol 25(1):67–71. https://doi.org/10.1017/S0265021507002591
Kernan S, Rehman S, Meyer T, Bourbeau J, Caron N, Tobias JD (2011) Effects of dexmedetomidine on oxygenation during one-lung ventilation for thoracic surgery in adults. J Minim Access Surg 7(4):227–231. https://doi.org/10.4103/0972-9941.85645
Kheirandish-Gozal L, Gozal D (2019) Obstructive sleep apnea and inflammation: proof of concept based on two illustrative cytokines. Int J Mol Sci 20(3):459. https://doi.org/10.3390/ijms20030459
Li Q, Zheng X (2017) Tumor necrosis factor alpha is a promising circulating biomarker for the development of obstructive sleep apnea syndrome: a meta-analysis. Oncotarget 8(16):27616–26. https://doi.org/10.18632/oncotarget.15203
Imani MM, Sadeghi M, Khazaie H, Emami M, Sadeghi Bahmani D, Brand S (2020) Evaluation of serum and plasma interleukin-6 levels in obstructive sleep apnea syndrome: a meta-analysis and meta-regression. Front Immunol 11:1343. https://doi.org/10.3389/fimmu.2020.01343
Sánchez-Pedrosa G, Vara Ameigeiras E, Casanova Barea J, Rancan L, Simón Adiego CM, Garutti Martínez I (2018) Role of surgical manipulation in lung inflammatory response in a model of lung resection surgery. Interact Cardiovasc Thorac Surg 27(6):870–877. https://doi.org/10.1093/icvts/ivy198
Meng L, Li L, Lu S, Li K, Su Z, Wang Y, Fan X, Li X, Zhao G (2018) The protective effect of dexmedetomidine on LPS-induced acute lung injury through the HMGB1-mediated TLR4/NF-κB and PI3K/Akt/mTOR pathways. Mol Immunol 94:7–17. https://doi.org/10.1016/j.molimm
Wu Y, Liu Y, Huang H, Zhu Y, Zhang Y, Lu F, Zhou C, Huang L, Li X, Zhou C (2013) Dexmedetomidine inhibits inflammatory reaction in lung tissues of septic rats by suppressing TLR4/NF-κB pathway. Mediators Inflamm 2013:562154. https://doi.org/10.1155/2013/562154
Ohta Y, Miyamoto K, Kawazoe Y, Yamamura H, Morimoto T (2020) Effect of dexmedetomidine on inflammation in patients with sepsis requiring mechanical ventilation: a sub-analysis of a multicenter randomized clinical trial. Crit Care 24(1):493. https://doi.org/10.1186/s13054-020-03207-8
Goodman RB, Pugin J, Lee JS, Matthay MA (2003) Cytokine-mediated inflammation in acute lung injury. Cytokine Growth Factor Rev 14(6):523–535. https://doi.org/10.1016/s1359-6101(03)00059-5
Ebert TJ, Hall JE, Barney JA, Uhrich TD, Colinco MD (2000) The effects of increasing plasma concentrations of dexmedetomidine in humans. Anesthesiology 93(2):382–394. https://doi.org/10.1097/00000542-200008000-00016
Acknowledgements
We gratefully acknowledge the participants and staff members at the General Hospital of Ningxia Medical University.
Funding
This work was supported by the National Natural Science Foundation of China (grant number: 81660198) and the Key Research and Development Program of Ningxia (grant number: 2021BEG03048).
Author information
Authors and Affiliations
Contributions
Na Li and Yonghai Zhang contributed to the design of the work, analysis of the data, and drafting of the manuscript. Fan Yang, Huiwen Zhang, Xiaoyang Yu, Xiaoyang Yu, Kaimei Lu and Jie Wang contributed to the acquisition, analysis, and interpretation of the data. Hanxiang Ma and Xinli Ni contributed to the project administration support and design of the work and wrote the original draft. All authors read and revised the manuscript and approved the final version.
Corresponding authors
Ethics declarations
Ethics approval
This study was approved by the Ethics Committee of the General Hospital of Ningxia Medical University (No. 2018–243) and conducted in accordance with the World Medical Association Declaration of Helsinki. All participants signed written informed consent forms.
Consent to participate
Informed consent was obtained from all individual participants included in the study or their guardians.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, N., Zhang, Y., Yang, F. et al. Effects of dexmedetomidine on oxygenation and inflammatory factors in patients undergoing uvulopalatopharyngoplasty: a prospective, randomized, placebo-controlled trial. Sleep Breath 27, 1099–1106 (2023). https://doi.org/10.1007/s11325-022-02711-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-022-02711-1