Abstract
Background and Objectives
Aspirin is a common drug for the treatment of pre-eclampsia. We aimed to explore whether quercetin as a supplement to aspirin could enhance the therapeutic outcome in pre-eclampsia rat models. We further aimed to evaluate the nucleotide-binding oligomerization domain-like receptor family, pyrin domain containing 3 (NLRP3) inflammasome as a potential pre-eclampsia-related molecular mechanism, which can be affected by quercetin treatment.
Methods
Rat pre-eclampsia models were established using an intravenous lipopolysaccharide injection after gestation. Rats were treated with aspirin and quercetin at 6–18 days after pregnancy. On day 20, blood, fetus, and placenta were harvested. Blood pressure and the level of proteinuria were measured every 4 days. Fetal outcomes were analyzed by pup body weight. Serum soluble Fms-like tyrosine kinase-1, PIGF, interleukin-6, and interleukin-10 levels were measured using the enzyme-linked immunosorbent assay. Caspase-1, NLRP3, apoptosis-associated speck-like protein containing a caspase recruitment domain, and p-caspase-1 levels in the placenta were assessed using western blot or quantitative real-time polymerase chain reaction analyses.
Results
Pre-eclampsia rat models showed a pronounced increase in systolic blood pressure and proteinuria after 4 days of pregnancy, while aspirin, quercetin, and aspirin/quercetin combinatory treatment significantly attenuated the blood pressure and proteinuria abnormalities. Notably, the aspirin/quercetin combinatory treatment showed the highest efficacy in attenuating pre-eclampsia-like symptoms. Placental caspase-1 and NLRP3 levels also showed the greatest attenuation in pre-eclampsia rats after aspirin/quercetin treatment.
Conclusions
Our data suggested that quercetin supplementation to aspirin is more effective in attenuating symptoms of pre-eclampsia and improving pregnancy outcomes compared with quercetin or aspirin alone. Quercetin can ameliorate placental NLRP3 inflammasome activation, which might serve as an underlying mechanism for its therapeutic efficacies in pre-eclampsia.
Similar content being viewed by others
Quercetin supplementation to aspirin, as a combinatory treatment, is more effective in attenuating symptoms of pre-eclampsia and improving pregnancy outcomes. | |
Quercetin can ameliorate placental nucleotide-binding oligomerization domain-like receptor family, pyrin domain containing 3 inflammasome activation, which contributes to the alleviation of pre-eclampsia. |
1 Introduction
Pre-eclampsia is a life-threatening condition that affects 2–8% of all pregnancies [1]. Characterized by disorders such as de novo hypertension, proteinuria, renal failure, neurological complications, increased liver enzymes, and uteroplacental dysfunction, pre-eclampsia is one of the major causes of fetal, maternal, and neonatal morbidity and mortality [2,3,4]. It is believed that inadequate placental vasculature caused by pre-eclampsia leads to episodes of hypoxia/reperfusion and impaired uteroplacental perfusion [5]. This is accompanied by the generation of cytokines, reactive oxygen species [6], lipid peroxidases, soluble Fms-like tyrosine kinase-1 (sFlt-1), and ET-1 [7], resulting in elevated inflammatory responses and exacerbation of the patient’s conditions [8].
The effective and safe pharmacological treatment of pre-eclampsia is still lacking. The complex etiology and high demand for safety profiles because of pregnancy are major hurdles for drug the development for pre-eclampsia. Currently, because of the anti-platelet and anti-inflammatory properties, low-dose aspirin is recommended for preventing or delaying the onset of pre-eclampsia [9, 10]. Unfortunately, recent studies suggest that the efficiency of low-dose aspirin in pre-eclampsia appears only to be modest or even non-responsive [11, 12] and high-dose aspirin is prohibited because of potential adverse effects such as birth defects [13]. This prompted researchers to pursue a new treatment regimen to improve the clinical management of pre-eclampsia [14,15,16]. Medical plants and vegetables have become a promising source for drug development [17], among which quercetin, a naturally occurring bioflavonoid derived from vegetables and plants such as apples, onions, and green tea, has recently stood out for its potency in regulating oxidative stress [18], vasodilation [19, 20], and inflammation [18, 21]. Our previous study has demonstrated that quercetin alone has similar efficacy to aspirin in treating pre-eclampsia in an Nω-nitro-l-arginine methyl ester hydrochloride (l-NAME)-induced pre-eclampsia rat model and quercetin supplementation appeared to exert a stronger efficacy than quercetin or aspirin alone [22].
As l-NAME and lipopolysaccharide (LPS) induction are two primary methods to establish pre-eclampsia models in rats, this study aims to explore if quercetin supplementation to aspirin could achieve greater therapeutic effects than aspirin in an LPS-induced pre-eclampsia rat model. We compared the combinatory treatment to quercetin or aspirin treatment alone in alleviating the symptoms of pre-eclampsia and reducing inflammatory cytokines and pre-eclampsia markers. Nucleotide-binding oligomerization domain-like receptor family, pyrin domain containing 3 (NLRP3) is one of the most well-studied inflammasomes in response to diverse stimuli [23]. Here, we aimed to investigate the role of inflammation in placenta tissues and how NLRP3 participates in such a process in pre-eclampsia. Our data could potentiate the application of quercetin as a supplement to aspirin in the clinical management of patients with pre-eclampsia.
2 Materials and Methods
2.1 Rat Pre-eclampsia Models and Treatment Procedures
Female Sprague–Dawley (SD) rats (Cyagen Biosciences Inc., Suzhou, China) with the body weight of 200–250 g were housed in a light-controlled and humidity-controlled room with free access to food and water. After a 1-week acclimatization, female SD rats were mated overnight with healthy male SD rats at a 2:1 ratio. Successful pregnancy was confirmed by the presence of vaginal spermatozoa. The date of pregnancy was designated as gestational day (GD) 1. Ten female rats (body weight of 200–250 g) with successful pregnancy were used for each group. Pregnant SD rats were randomly divided into five groups, which received the following interventions: (1) control group, which received no interventions; (2) pre-eclampsia (PE) group, which were intravenously injected with 1.0 µg/kg body weight LPS (Sigma-Aldrich, St. Louis, MO, USA) at GD5 (to create pre-eclampsia models, i.e., pre-eclampsia rats), (3) pre-eclampsia plus quercetin (PE+Que) group, in which pre-eclampsia rats received quercetin treatment only at the dose of 2 mg/kg body weight via an intraperitoneal injection; (4) pre-eclampsia plus aspirin (PE+Asp) group, in which pre-eclampsia rats received aspirin treatment only at the dose of 1.5 mg/kg body weight. Aspirin was given mixed with rodent dough (1 g, Transgenic Dough DietBacon Flavor; Bio-Serv, Flemington, NJ, USA); and (5) pre-eclampsia plus quercetin plus aspririn (PE+Que+Asp) group, in which pre-eclampsia rats received both quercetin and aspirin treatments. Aspirin and quercetin were given at GD6–18. Rats were sacrificed using pentobarbital sodium anesthesia on day 20 and the number of visible and absorbed pups was recorded. Fetal placental tissue samples and serum were collected. The animal study was approved by the Ethics Committee of Cangzhou Central Hospital.
2.2 Western Blot and ELISA and Measurement of Proteinuria and Blood Pressure
Western blot was performed using placental tissue, which was homogenized and lysed using the RIPA buffer. Then, 50 μg of protein were loaded in each lane and SDS gels of 10–14% were used. Proteins were transferred to nitrocellulose membranes and blocked with 1% bovine serum albumin in phosphate-buffered saline (pH 7.4). All antibodies were acquired from Cell Signaling Technologies (Danvers, MA, USA) and used according to the recommendations of the vendors. Placental tissue sample was homogenized in 0.2% H3PO4 solution and then centrifuged for 10 min at 3500g at 4 °C to collect the supernatant before the enzyme-linked immunosorbent assay (ELISA). The ELISA kits were acquired from Abcam (Cambridge, MA, USA). The Coomassie brilliant blue kit (Jiancheng Institute of Biotechnology, Nanjing, China) was employed to detect proteinuria under the instructions from the manufacturer. Blood pressure was measured through a noninvasive tail-cuff method using the BP-2000 Blood Pressure Analysis System (Visitech Systems, Inc., Apex, NC, USA). Systolic blood pressure was assessed with five continuous values with variations < 6 mmHg averaged to define maternal systolic blood pressure.
2.3 Quantitative Real-Time PCR
Quantitative real-time polymerase chain reaction (PCR) was performed after total RNA extraction using the TRIZOL agent. The following primers were used. sFlt-1: 5′-AAGGTCTACAGCACCAAG-3′ (F); 5′-CACATCATCAGAGCTTCC-3′ (R); PIGF: forward CTCAGGATGTACTCTGCGAATG, reverse TTTGCTTCCTCTTCCCCTTG; interleukin (IL)-6: forward 5′-CAAAGCCAGAGTCATTCAGA-3′, reverse 5′-TTGCCGAGTAGACCTCATAGT-3′; IL-10: forward 5′-TCTACAAGGCCATGAATGAG-3′, reverse 5′-GAGAGAGGTACAAACGAGG-3′; NLRP3: forward 5′-CCTGTGTGGGAACAAGTATGC-3′, reverse 5′-TCCCATCAGGTCACCAAGAG-3; Caspase-1: forward 5′-CGAGTGGTTCCCTCAAGTTT-3′, reverse 5′-TGAAAGTCTGTGCTGCAGAT-3′; β-actin: forward 5′-AGGGAAATCGTGCGTGACAT-3′, reverse 5′-GAACCGCTCATTGCCGATAG-3′.
2.4 Statistical Analysis
All experiments were performed using at least three independent replicates for each group. One/two-way analysis of variance with an appropriate post hoc test was used for comparisons, and p < 0.05 was used as the criterion for statistical significance. GraphPad prism was used to perform the analysis.
3 Results
3.1 Quercetin/Aspirin Treatments Attenuate Hypertension and Proteinuria in Pre-eclampsia Rat Models
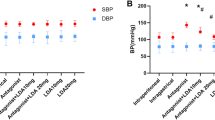
We constructed pre-eclampsia rat models by intravenously injecting a low dose of LPS (1 μg/kg body weight) into pregnant rats. As shown in Fig. 1, following the LPS injection at GD5, systolic blood pressure (Fig. 1A) and proteinuria (Fig. 1B) demonstrated an immediate elevation, while healthy controls showed stable levels of systolic pressure and proteinuria levels. Rats were then treated with quercetin (2 mg/kg), aspirin (1.5 mg/kg), or both since GD6. Quercetin and aspirin alone both showed significant effects in attenuating the levels of systolic pressure and proteinuria, but treating with a combination of quercetin and aspirin led to the most pronounced effects. This data indicated that quercetin and aspirin are effective in reducing hypertension and proteinuria, which are pre-eclampsia-like symptoms, and the combination of quercetin and aspirin outperformed either drug alone.
Quercetin (Que) supplement to aspirin (Asp) attenuated systolic blood pressure and proteinuria in lipopolysaccharide-induced pre-eclampsia (PE)-like rats. A Systolic blood pressure of each indicated group was measured non-invasively using the tail-cuff method. B The 24-h proteinuria in each group was detected using Coomassie brilliant blue kits. Data were shown with mean ± standard deviation. Ten rats were used for each group. *p < 0.05, **p < 0.01, and ***p < 0.001 compared to the PE group. ^p < 0.05 compared to the PE+Asp group. Two-way analysis of variance followed by the Tukey’s multiple comparisons test. GD gestational day
3.2 Quercetin/Aspirin Treatments Improve Pregnancy Outcomes in Pre-eclampsia Rat Models
We next assessed if the attenuation of hypertension and proteinuria translated to better gestational outcomes measured by pup body length and weight at GD18. As shown in Fig. 2, while pre-eclampsia rats showed a dramatic reduction in both pup body length and weight, quercetin, aspirin, and the combinatory treatment of quercetin and aspirin resulted in higher pup body length (Fig. 2A) and weight (Fig. 2B). The most significant improvement of gestational outcome was also seen for pre-eclampsia rats treated with both quercetin and aspirin.
Quercetin (Que) supplement to aspirin (Asp) ameliorated pregnancy outcomes in lipopolysaccharide-induced pre-eclampsia (PE)-like rats. Pups body length (A) and weight (B) were compared. N = 83 for control, 69 for PE, 72 for PE+Que, 74 for PE+Asp, and 79 for PE+Que+Asp. Data were shown with mean ± standard deviation. *p < 0.05, **p < 0.01, and ***p < 0.001 compared to the PE group. ^^p < 0.01 compared to the PE+Asp group. One-way analysis of variance followed by the Dunnett’s T3 multiple comparisons test
3.3 sFlt-1/PIGF Balance was Restored by Quercetin/Aspirin Treatments
As the imbalance of sFlt-1/PIGF is one of the characteristics and molecular underpinnings of pre-eclampsia, we investigated if the alleviation of pre-eclampsia symptoms and improved gestational outcomes could be attributed to the role of quercetin and aspirin combinatory treatment in restoring the sFlt-1/PIGF balance. As shown in Fig. 3, pre-eclampsia rats were characteristic of upregulated sFlt-1 and downregulated PIGF in serum. Treatment with quercetin, aspirin, or the combination of quercetin and aspirin expectedly led to a reduction in serum sFlt-1 and an increase in PIGF, with combinatory treatment showing the highest effects in normalizing these levels (Fig. 3A,B). The sFlt-1/PIGF ratios in both serum (Fig. 3C) and placental tissue (Fig. 3D) were also lowered most prominently by quercetin and aspirin combinatory treatment.
Quercetin (Que) supplement to aspirin (Asp) ameliorated soluble Fms-like tyrosine kinase-1 (sFlt-1)/PIGF balance in lipopolysaccharide-induced pre-eclampsia (PE)-like rats. The enzyme-linked immunosorbent assay was used to measure the serum sFlt-1 (A), PIGF (B) levels and their ratio (C) on gestational day 20. D Quantitative reverse transcription-polymerase chain reaction was used to measure the mRNA expressions of sFlt-1 and PIGF in placenta from different groups. Data were shown with mean ± standard deviation. Eight rats were used for each group in the enzyme-linked immunosorbent assay. Ten placental homogenates were mixed for each group and the quantitative reverse transcription-polymerase chain reaction was repeated four times. *p < 0.05, **p < 0.01, and ***p < 0.001 compared to the PE group. ^^p < 0.01 compared to the PE+Asp group. One-way analysis of variance followed by the Dunnett’s T3 multiple comparisons test
3.4 Quercetin/Aspirin Treatments Alleviate Placental Inflammation in Pre-eclampsia Rat Models
Another pathological signature of pre-eclampsia is placental inflammation and we therefore investigated the potency of quercetin/aspirin treatments in alleviating placental inflammation in pre-eclampsia rats. The ELISA (Fig. 4A,B) and quantitative reverse transcription-PCR (Fig. 4C,D) analyses of IL-6 and IL-10 suggested that the expression of IL-6, a pro-inflammatory cytokine was reduced while IL-10, an anti-inflammatory cytokine, was elevated after quercetin and aspirin treatments.
Quercetin (Que) supplement to aspirin (Asp) ameliorated placental inflammatory responses in lipopolysaccharide-induced pre-eclampsia (PE)-like rats. The enzyme-linked immunosorbent assay was used to measure the placental interleukin (IL)-6 (A) and IL-10 (B) levels on gestational day 20. Quantitative reverse transcription-polymerase chain reaction was used to measure the mRNA expressions of IL-6 (C) and IL-10 (D) in the placenta from different groups. Data were shown with mean ± standard deviation. Eight were used for each group in the enzyme-linked immunosorbent assay. Ten placental homogenates were mixed for each group and the quantitative reverse transcription-polymerase chain reaction was repeated four times. *p < 0.05, **p < 0.01, and ***p < 0.001 compared to the PE group. ^p < 0.05 and ^^p < 0.01 compared to the PE+Asp group. One-way analysis of variance followed by the Dunnett’s T3 multiple comparisons test
3.5 Quercetin/Aspirin Treatments Ameliorate Placental NLRP3 Inflammasome Activation in Pre-eclampsia Rat Models
To elucidate the molecular mechanism of the effects of quercetin in treating pre-eclampsia, important members of the NLRP3 inflammasome, including caspase-1, NLRP3, pro-caspase-1, and apoptosis-associated speck-like protein containing a caspase recruitment domain, in placental tissues was analyzed by quantitative real-time PCR and western blot. Our data indicated that quercetin and aspirin effectively downregulated caspase-1, NLRP3, apoptosis-associated speck-like protein containing a caspase recruitment domain, and pro-caspase-1 (Fig. 5A–G) on both mRNA and protein levels.
Quercetin (Que) supplement to aspirin (Asp) ameliorated placental nucleotide-binding oligomerization domain-like receptor family, pyrin domain containing 3 (NLRP3) inflammasome activation in lipopolysaccharide-induced pre-eclampsia (PE)-like rats. Quantitative reverse transcription-polymerase chain reaction was used to measure the mRNA expressions of caspase-1 (A) and NLRP3 (B) in the placenta from different groups on gestational day 20. C Western blotting was used to measure placental protein expressions of NLRP3, apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC), pro-caspase-1, and caspase-1 from different groups on gestational day 20. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as a loading control and the expressions were normalized to control group (D–G). Data were shown with mean ± standard deviation. Ten placental homogenates were mixed for each group and the Western blot, Quantitative reverse transcription-polymerase chain reaction was repeated for four times. *p < 0.05, **p < 0.01, and ***p < 0.001 compared to the PE group. ^p < 0.05 and ^^p < 0.01 compared to the PE+Asp group. One-way analysis of variance followed by the Dunnett’s T3 multiple comparisons test
4 Discussion
Here, we investigate the effects of quercetin supplementation to aspirin as a new treatment regimen to alleviate symptoms and improve gestational outcomes of rats with pre-eclampsia-like symptoms. The potential adverse effects associated with aspirin, which prohibits the use of a high dose of aspirin in pregnant women, as well as recent reports of suboptimal efficacies of aspirin in treating pre-eclampsia prompted us to investigate if supplementation with another biocompatible drug while achieving similar or even higher therapeutic effects can improve therapeutic efficacies. During pre-eclampsia development, inflammation intensifies, along with elevated lipid peroxidation products, reactive oxygen species, and anti-angiogenic factors including sFlt-1 and vascular endothelial growth factor in placental tissue and maternal circulation [24]. Quercetin as a lead flavonoid in clinical development has garnered significant attention predominantly owing to its safety profile and strong anti-inflammatory efficacies. Prior studies support that flavonoid supplementation to aspirin is efficacious in stimulating platelet aggregation [25], rendering enhanced antithrombotic effects. It is also recently reported that a potential interaction between quercetin and aspirin exists, thereby exerting a synergic, rather than additive, effect [25] that makes this treatment regimen attractive. In an L-NAME-induced pre-eclampsia rat model, we showed that quercetin supplementation to aspirin indeed achieved higher effects of lowering systolic blood pressure, proteinuria, as well as improving gestational outcomes [22]. The combinatory treatment also achieved a stronger effect in suppressing lipid peroxidation products. Inflammatory responses, measured by the production of IL-6 and IL-10, were also suppressed [22]. Our data further confirmed the utility of such combinatory treatment in the LPS-induced PE rat model, which tested the efficacy of this combinatory approach in another animal model of pre-eclampsia, serving as important evidence that supports further development of such combinatory treatment as a clinical tool in managing pre-eclampsia.
One shining point of our study is that we also focused on investigating how the NLRP3 inflammasome was affected by quercetin/aspirin combinatory treatment, which has been rarely explored previously. NLRP3 is one of the most well-studied inflammasomes that in response to diverse stimuli binds to the inactive pro-caspase-1 enzyme and apoptosis-associated speck-like protein containing a caspase recruitment domain, forming an inflammasome complex that further promotes pro-inflammatory cytokine production [23]. Yet there is scant information on the role NLRP3 inflammasome plays in pre-eclampsia, and the interaction between quercetin and NLRP3 has been demonstrated in studying neurotoxicity [26]. It was recently demonstrated that expression of the NLRP3 inflammasome is pronouncedly higher in women with pre-eclampsia compared with normotensive pregnant women. The fact that quercetin/aspirin treatment greatly downregulates the expression of the NLRP3 inflammasome is consistent with attenuated cytokine production in pre-eclampsia rats. Our study also corroborated that quercetin may also serve as a new potent inhibitor of the NLRP3 inflammasome.
Despite demonstrating the remarkable efficacy of the quercetin/aspirin treatment in rat models, further studies that gauge the efficacy in human patients are extremely important. As both quercetin and aspirin have been used in humans, we foresee a shorter clinical path. Further, our study did not evaluate the effects of dose and treatment schedule on gestational outcomes, which needs to be addressed to facilitate translation.
5 Conclusions
In summary, we have shown that in rats with LPS-induced pre-eclampsia, a quercetin supplement to aspirin leads to greater efficacies in alleviating pre-eclampsia-like symptoms including elevated systolic blood pressure and proteinuria, as well as improving gestational outcomes. Quercetin was also shown to synergize with aspirin to inhibit the NLRP3 inflammasome, which is a potential underlying mechanism in its anti-inflammatory effects. These data support the development of quercetin supplementation to aspirin as a new strategy for the therapy of pre-eclampsia in humans.
References
Giannakou K, Evangelou E, Papatheodorou SI. Genetic and non-genetic risk factors for pre-eclampsia: umbrella review of systematic reviews and meta-analyses of observational studies. Ultrasound Obstet Gynecol. 2018;51:720–30. https://doi.org/10.1002/uog.18959.
Nicholson WK, Stones W, Visser GHA, Barnea ER, Nassar AH, Federation International Gynecology and Obstetrics Safe Motherhood and Newborn Health Committee. Don’t forget eclampsia in the efforts to reduce maternal morbidity and mortality. Int J Gynaecol Obstet. 2021;152:165–71. https://doi.org/10.1002/ijgo.13530.
Xiao Z, Wang Y, Thai PN, Li X, Lu X, Pu J. Mechanisms linking hyperglycemia in pregnancy to the offspring cardiovascular system dysfunction. STEMedicine. 2021;2: e91. https://doi.org/10.37175/stemedicine.v2i7.91.
Xiao S, Zhang Q, Zhang M, Hu R, Liu R. A modified Mediterranean diet against gestational diabetes mellitus. STEMedicine. 2022;3: e129. https://doi.org/10.37175/stemedicine.v3i3.129.
Phipps EA, Thadhani R, Benzing T, Karumanchi SA. Pre-eclampsia: pathogenesis, novel diagnostics and therapies. Nat Rev Nephrol. 2019;15:386. https://doi.org/10.1038/s41581-019-0156-1.
Williamson RD, McCarthy FP, Khashan AS, Totorika A, Kenny LC, McCarthy C. Exploring the role of mitochondrial dysfunction in the pathophysiology of pre-eclampsia. Pregnancy Hypertens. 2018;13:248–53. https://doi.org/10.1016/j.preghy.2018.06.012.
Graupner O, Karge A, Flechsenhar S, Seiler A, Haller B, Ortiz JU, et al. Role of sFlt-1/PlGF ratio and feto-maternal Doppler for the prediction of adverse perinatal outcome in late-onset pre-eclampsia. Arch Gynecol Obstet. 2020;301:375–85. https://doi.org/10.1007/s00404-019-05365-9.
Stodle GS, Silva GB, Tangeras LH, Gierman LM, Nervik I, Dahlberg UE, et al. Placental inflammation in pre-eclampsia by Nod-like receptor protein (NLRP)3 inflammasome activation in trophoblasts. Clin Exp Immunol. 2018;193:84–94. https://doi.org/10.1111/cei.13130.
Crandon AJ, Isherwood DM. Effect of aspirin on incidence of pre-eclampsia. Lancet. 1979;1:1356. https://doi.org/10.1016/s0140-6736(79)91996-2.
Goodlin RC, Haesslein HO, Fleming J. Aspirin for the treatment of recurrent toxaemia. Lancet. 1978;2:51. https://doi.org/10.1016/s0140-6736(78)91367-3.
Navaratnam K, Alfirevic A, Jorgensen A, Alfirevic Z. Aspirin non-responsiveness in pregnant women at high-risk of pre-eclampsia. Eur J Obstet Gynecol Reprod Biol. 2018;221:144–50. https://doi.org/10.1016/j.ejogrb.2017.12.052.
Van Doorn R, Mukhtarova N, Flyke IP, Lasarev M, Kim K, Hennekens CH, et al. Dose of aspirin to prevent preterm preeclampsia in women with moderate or high-risk factors: a systematic review and meta-analysis. PLoS One. 2021;16: e0247782. https://doi.org/10.1371/journal.pone.0247782.
Gupta U, Cook JC, Tassinari MS, Hurtt ME. Comparison of developmental toxicology of aspirin, (acetylsalicylic acid) in rats using selected dosing paradigms. Birth Defects Res Dev Reprod Toxicol. 2003;68:27–37. https://doi.org/10.1002/bdrb.10007.
Li QH, Yin LX, Si YR, Zhang CJ, Meng YH, Yang WW. The bioflavonoid quercetin improves pathophysiology in a rat model of preeclampsia. Biomed Pharmacother. 2020;127: 110122. https://doi.org/10.1016/j.biopha.2020.110122.
Amaral LM, Wallace K, Owens M, LaMarca B. Pathophysiology and current clinical management of preeclampsia. Curr Hypertens Rep. 2017;19:61. https://doi.org/10.1007/s11906-017-0757-7.
Xie J, Zhang AH, Sun H, Yan GL, Wang XJ. Recent advances and effective strategies in the discovery and applications of natural products. RSC Adv. 2018;8:812–24. https://doi.org/10.1039/c7ra09475b.
Thomford NE, Senthebane DA, Rowe A, Munro D, Seele P, Maroyi A, et al. Natural products for drug discovery in the 21st century: innovations for novel drug discovery. Int J Mol Sci. 2018;19: 1578. https://doi.org/10.3390/ijms19061578.
Lesjak M, Beara I, Simin N, Pintac D, Majkic T, Bekvalac K, et al. Antioxidant and anti-inflammatory activities of quercetin and its derivatives. J Funct Foods. 2018;40:68–75. https://doi.org/10.1016/j.jff.2017.10.047.
Duarte J, Perezvizcaino F, Zarzuelo A, Jimenez J, Tamargo J. Vasodilator effects of quercetin in isolated rat vascular smooth-muscle. Eur J Pharmacol. 1993;239:1–7. https://doi.org/10.1016/0014-2999(93)90968-N.
Luna-Vazquez FJ, Ibarra-Alvarado C, Rojas-Molina A, Rojas-Molina I, Zavala-Sanchez MA. Vasodilator compounds derived from plants and their mechanisms of action. Molecules. 2013;18:5814–57. https://doi.org/10.3390/molecules18055814.
Li Y, Yao JY, Han CY, Yang JX, Chaudhry MT, Wang SN, Liu HN, Yin YL. Quercetin, inflammation and immunity. Nutrients. 2016;8:167. https://doi.org/10.3390/nu8030167.
Yang SY, Song LL, Shi XF, Zhao N, Ma YX. Ameliorative effects of pre-eclampsia by quercetin supplement to aspirin in a rat model induced by l-NAME. Biomed Pharmacother. 2019;116: 108969. https://doi.org/10.1016/j.biopha.2019.108969.
Jo EK, Kim JK, Shin DM, Sasakawa C. Molecular mechanisms regulating NLRP3 inflammasome activation. Cell Mol Immunol. 2016;13:148–59. https://doi.org/10.1038/cmi.2015.95.
Szarka A, Rigo J Jr, Lazar L, Beko G, Molvarec A. Circulating cytokines, chemokines and adhesion molecules in normal pregnancy and preeclampsia determined by multiplex suspension array. BMC Immunol. 2010;11:59. https://doi.org/10.1186/1471-2172-11-59.
Stainer AR, Sasikumar P, Bye AP, Unsworth AJ, Holbrook LM, Tindall M, et al. The metabolites of the dietary flavonoid quercetin possess potent antithrombotic activity, and interact with aspirin to enhance antiplatelet effects. TH Open. 2019;3:e244–58. https://doi.org/10.1055/s-0039-1694028.
Han XJ, Xu TS, Fang QJ, Zhang HJ, Yue LJ, Hu G, et al. Quercetin hinders microglial activation to alleviate neurotoxicity via the interplay between NLRP3 inflammasome and mitophagy. Redox Biol. 2021;44: 1347. https://doi.org/10.1016/j.redox.2021.102010.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
No funding was received for the preparation of this article.
Conflict of Interest
The authors have no conflicts of interest that are directly relevant to the content of this article.
Ethics Approval
The animal study was approved by the Ethics Committee of Cangzhou Central Hospital.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Availability of Data and Material
Data and materials could be obtained upon request to the corresponding author.
Code Availability
Not applicable.
Author Contributions
SY, JZ, DC, JD, YZ, and LS conducted the experiments, analyzed the data, and wrote the manuscript; SY and JZ conceived the study, reviewed the data, and revised the manuscript.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Yang, S., Zhang, J., Chen, D. et al. Quercetin Supplement to Aspirin Attenuates Lipopolysaccharide-Induced Pre-eclampsia-Like Impairments in Rats Through the NLRP3 Inflammasome. Drugs R D 22, 271–279 (2022). https://doi.org/10.1007/s40268-022-00402-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40268-022-00402-6